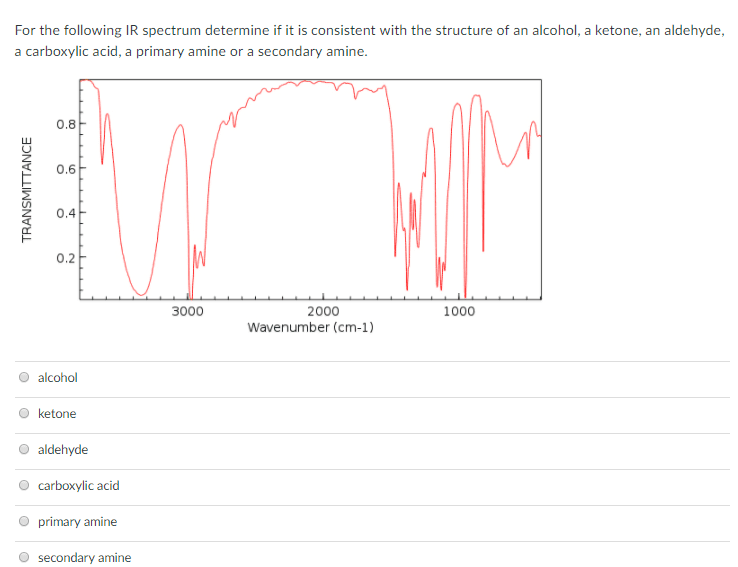
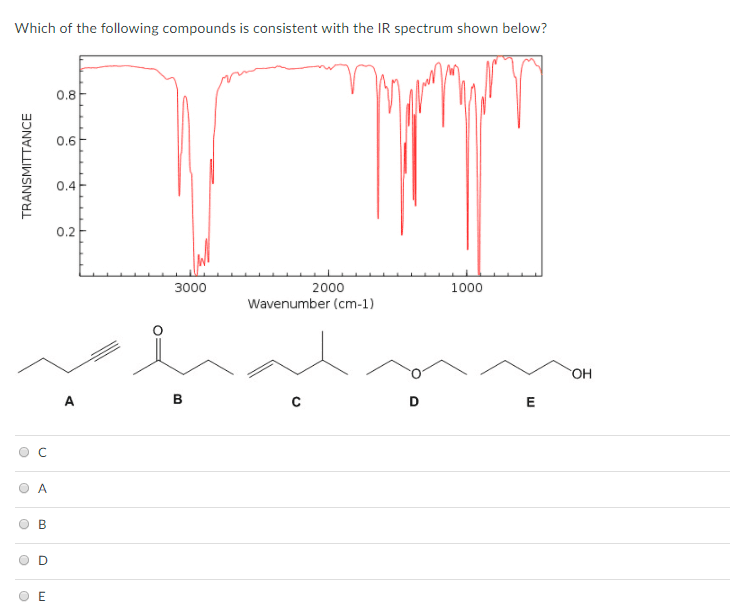
An acetal forms when two alcohol molecules react with an aldehyde or a ketone. The process involves the addition of two âOR groups from the alcohol to the carbonyl group, and the elimination of a water molecule. R represents the hydrocarbon chain of the alcohol. The general reaction and structure for an acetal are shown here.
Part A: Draw the acetal produced when ethanol adds to ethanal.
Part B: Draw the acetal produced when ethanol adds to propanone.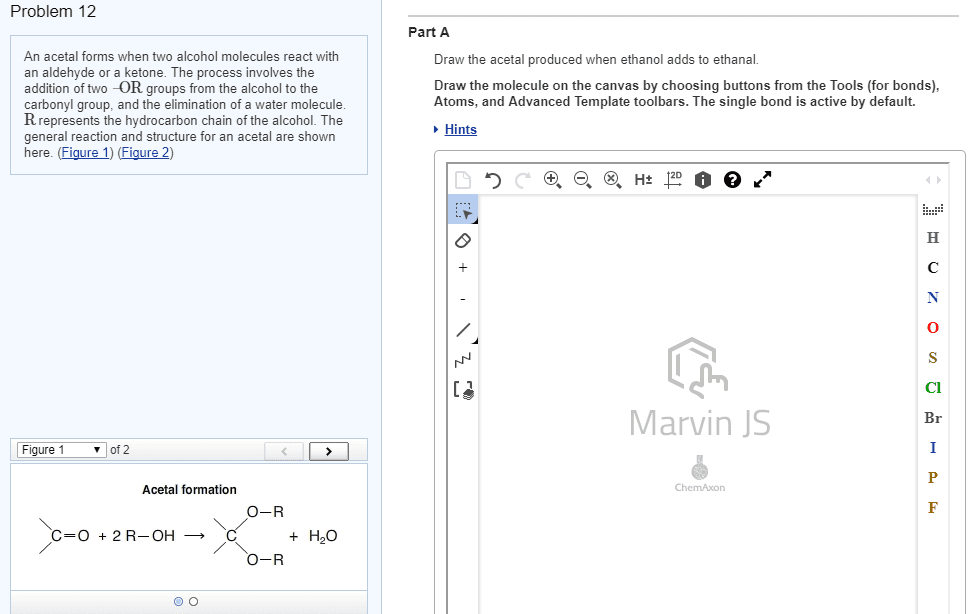
Problem 12 Part A An acetal forms when two alcohol molecules react with an aldehyde or a ketone. The process involves the addition of two -OR groups from the alcohol to the carbonyl group, and the elimination of a water molecule R represents the hydrocarbon chain of the alcohol. The general reaction and structure for an acetal are shown here. (Figure 1) (Figure 2) Draw the acetal produced when ethanol adds to ethanal. Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. The single bond is active by default. Hints Cl Marvin JS Figure 1 â¼ of 2 Acetal formation O-R O-R