1. Complete and balance the following chemical reaction: NH4NO3(s) + H2O(l) --> ?
Given
2. Volume of DI water used 50 mL
3. Mass of DI water 50 g
4. Initial temperature of water, Ti 22.5°C
5. Mass of NH4NO3 3.0 g
6. Moles of NH4NO3 ________0.0375__________mol
7. Final temperature of water, Tf 18.9°C
8. change in temperature of the water (?T) -3.6 °C
9. calculate the heat associate with water, qH2O ___ ___-752.4_____________ J
m*c* deltaT = (50g)(4.184 J/g*k)(-3.6) = -752.4
10. the heat either released or absorbed by the reaction (?Hrxn) ________+752.4____________ J
Question: [Please notice the units]
11. Calculate the change in enthalpy (?Hrxn) for this reaction. _____________ kJ ??
12. Calculate the heat of reaction in kJ per mole of NH4NO3 ______________ kJ/mol ??
13. If the heat capacity of the calorimeter was 25 J/oC, what would the new change in enthalpy (?Hrxn) for this reaction be? (Use equation 7 to recalculate the value from #10) ____________________ kJ/mol ?? [Equation 7: - q(rxn) = q(soln) + q(calorimeter)
14. Is this an exo- or endothermic reaction? Explain the reasoning behind your choice. ??
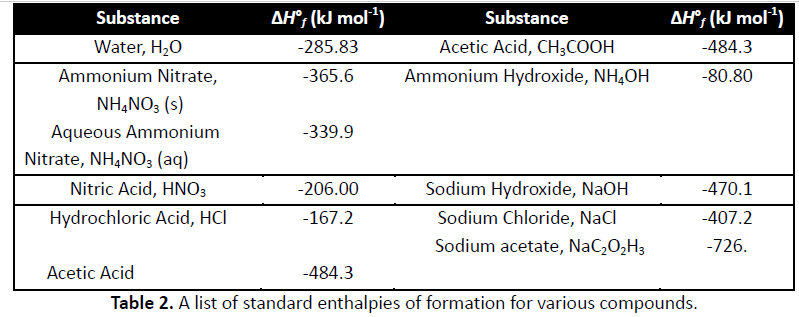
15. Calculate the change in enthalpy for the reaction (?Hrxn) per mole using equation 8 and Table 2. NH4NO3 (s) --> NH4NO3 (aq)
__________________kJ/mol ?? [Equation 8: ?Hrxn = sum{ n?Hf (products) - sum{ n?Hf (reactants)
16. What is the percent error between your value and the value calculated from 13? What are some possible sources of error in the experiment?
__________________% error
Substance Water, H20 Ammonium Nitrate, NHANO3 (s) Aqueous Ammonium AH, (kJ mol) 484.3 80.80 Substance f (kJ mol 285.83 365.6 Acetic Acid, CH3COOH Ammonium Hydroxide, NH4 OH 339.9 Nitrate, NH4NOs (aq) 470.1 -407.2 -726 Sodium Hydroxide, NaOH Nitric Acid, HNO3 Hydrochloric Acid, HCI -206.00 167.2 Sodium Chloride, NaCl Sodium acetate, NaC202H3 Acetic Acid 484.3 Table 2. A list of standard enthalpies of formation for various compounds


