CHM 2123 Lecture 2: Lab2-TheKineticStudyofNucleophilicSubstitution
Document Summary
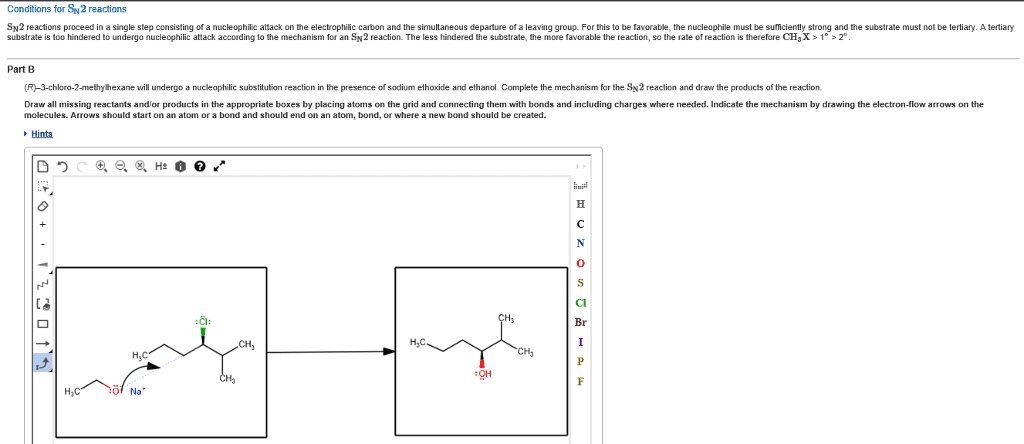
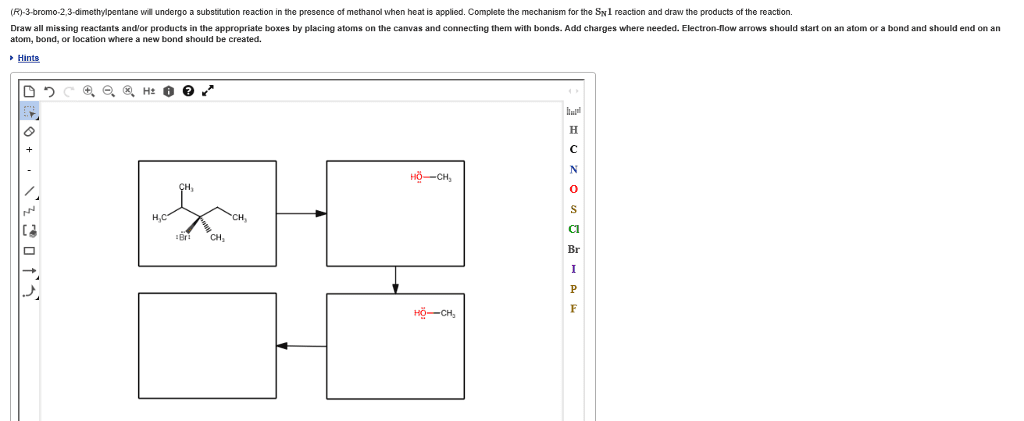
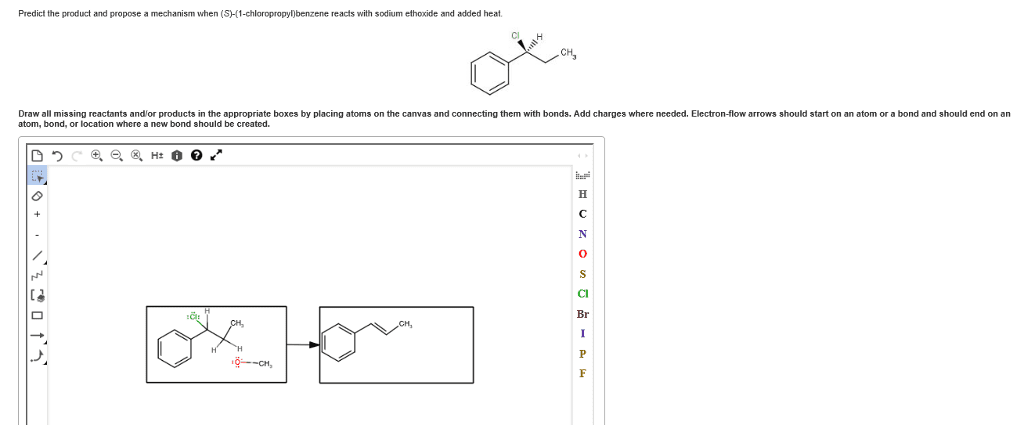
The purpose of the experiment in part a was to determine the effect of a leaving group on the rate of a nucleophilic substitution reaction. Two leaving groups were examined, chlorine and bromine. Substitution reactions involve a nucleophile (a species that bears a partial or formal negative charge) and an electrophile (which bears a partial or formal positive charge), which is usually attached to a leaving group. There are two major classes of substitution reactions and they both differ in their mechanisms. In part a it is the s n 2 reaction occurring. The s n 2 reaction happens in one bimolecular step, so it is a concerted mechanism. The nucleophile approaches from the back at an angle of 180 o to the leaving group, this results in an inversion of configuration. This inversion is characteristic of the s n 2 reaction and so therefore will produce products that are stereospecific.