CHM 2120 Lecture Notes - Lecture 14: Elimination Reaction, Cyclohexane Conformation, Nucleophile
Document Summary
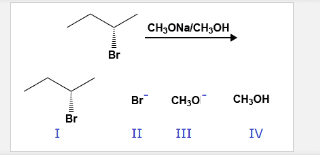
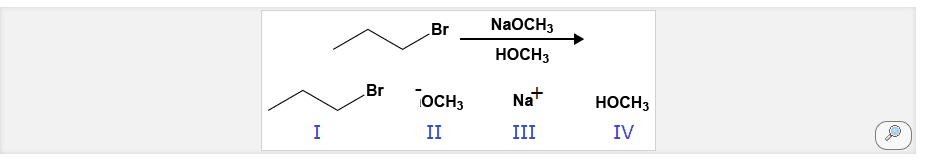
2 steps: leaving group leaves (rds), and carbocation forms (tertiary is most stable carbocation, base attacks neighboring hydrogen and this leaves a double bond to form between the alpha and beta carbons. Alkyl chain: tertiary preferred, with beta carbon being the more substituted one. Base: solvent may act as base, weak base so it waits for leaving group to leave on its own. So, if a base is weaker than the leaving group then the leaving group then type 1 happens because it is not strong enough to forcefully kick out the leaving group. There will always be competition between sn1 and e1. The beta carbon is the carbon closest to the alpha, we need to get most substituted alkene. Zaitse(cid:448)"s rule: the most substituted the alkene the more stable that alkene will be. There can be a cis, trans disubstituted alkenes. Tetrasubstituted is the most stable alkene that can form.