CHM 1311 Lecture Notes - Lecture 5: Kinetic Energy, Heat Capacity, Calorimetry
Document Summary
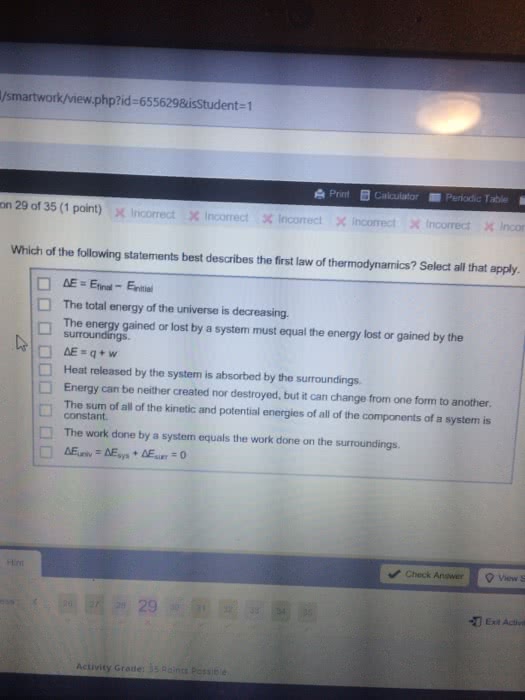
12:17 pm: the internal energy of a system is the sum of the kinetic and potential energies in the system. For atoms and molecules in chemical reactions: kinetic energy molecular or atomic motion, movement of electrons, potential energy attractive or repulsive interactions between electrons, nuclei, atoms, and molecules. U = change in internal energy q = heat energy transferred. Sign of q: positive - heat absorbed by system, negative - heat absorbed by surroundings w= work done on or by system. Sign of w: positive - work done on system, negative - work done by system. 1 g of water must absorb 4. 18 j of energy (in the form of heat) to increase the temperature by 1oc. Determining heat change (q) heat change = q = (c)(mass)( t) The form in which it naturally exists at 1 bar and at 25 c (298 k: calorimetry, hess" law, heats of formation, looking at bond energies (estimate)