CHEM 154 Lecture Notes - Lecture 24: Reactive Intermediate, Toothpaste, Hydrogen Peroxide
90 views4 pages
Document Summary
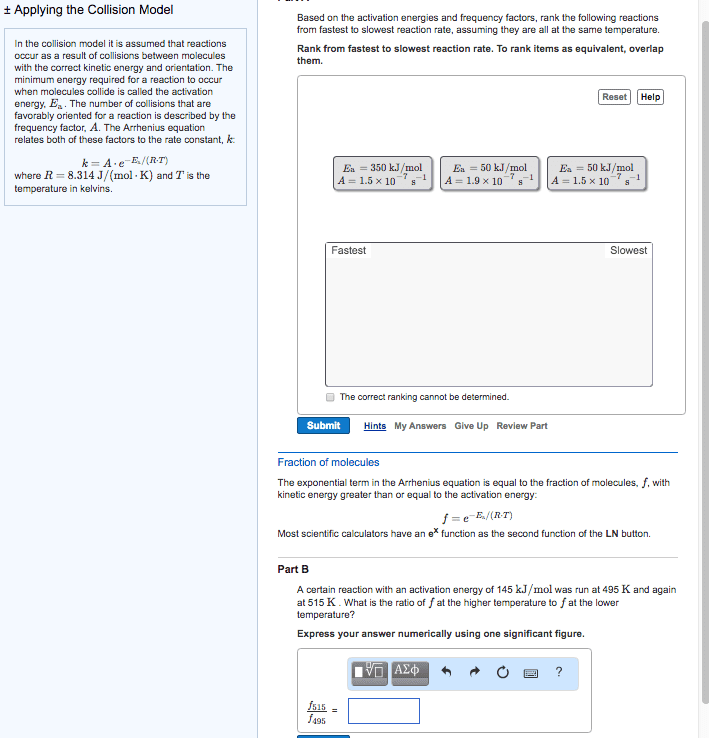
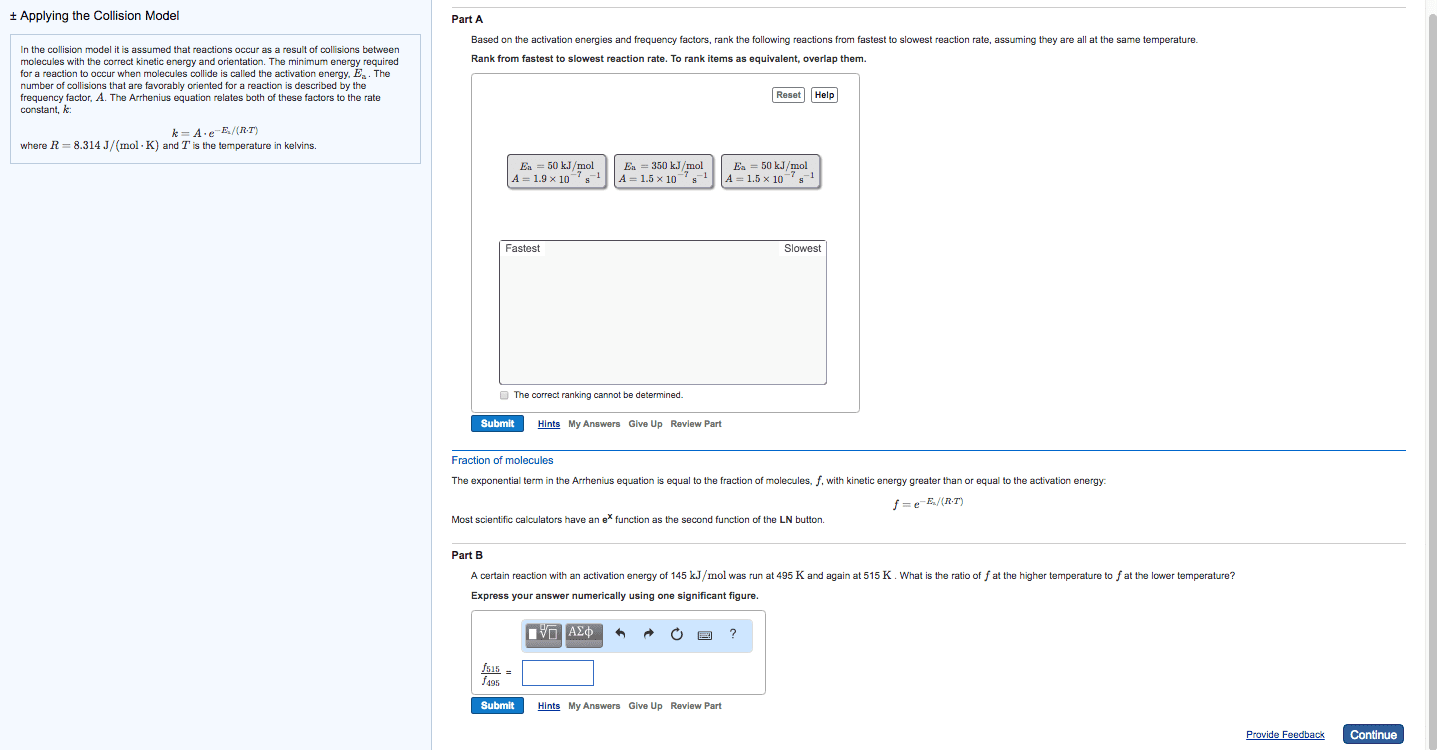
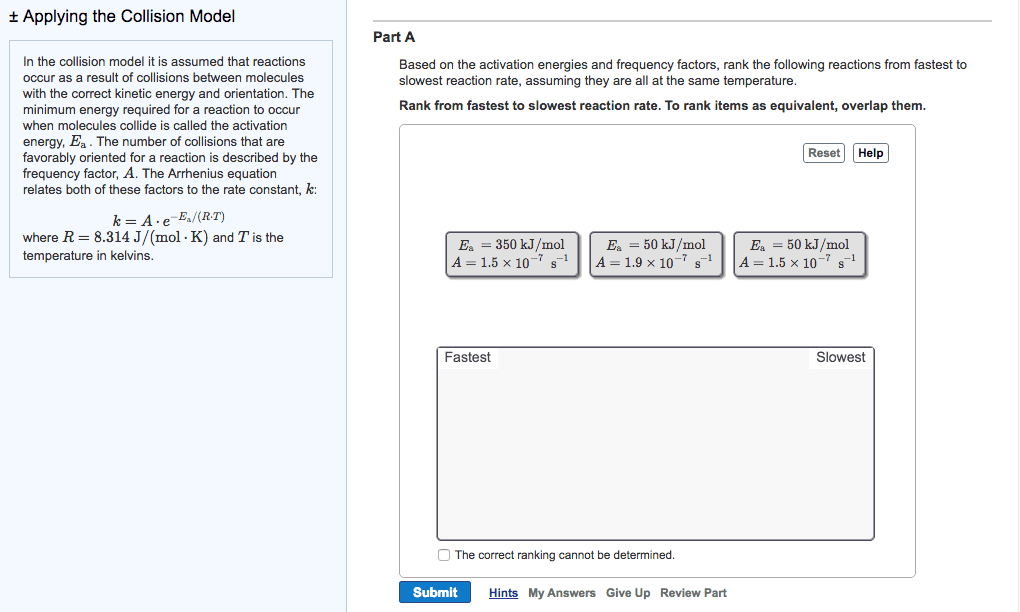
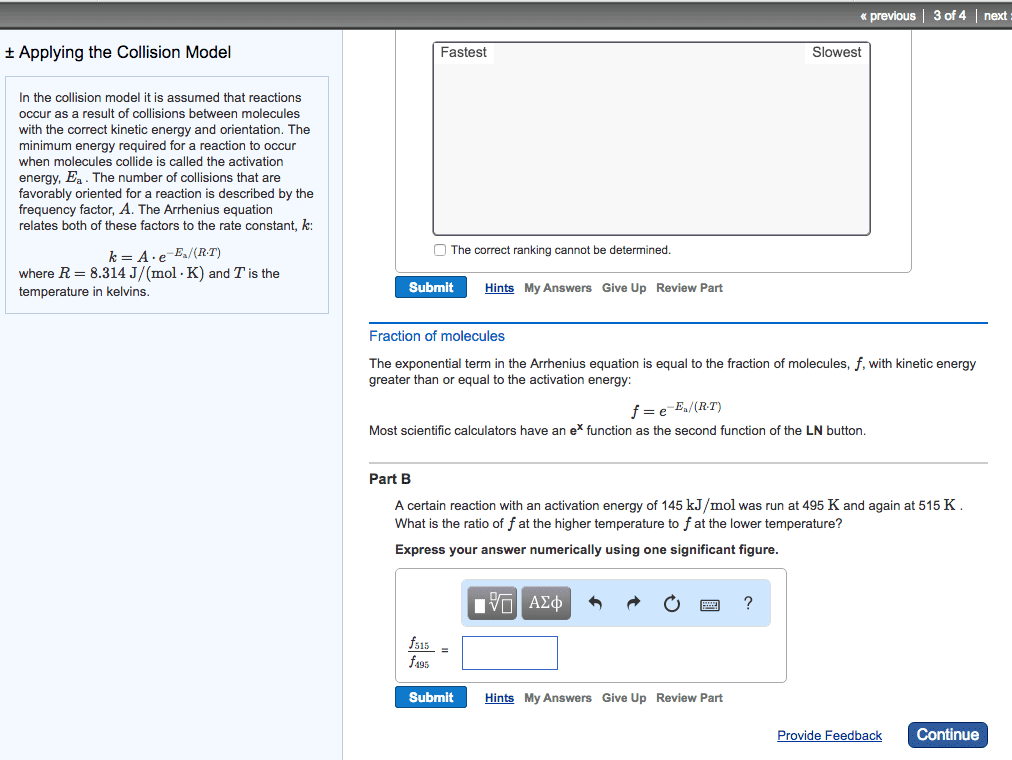
The half-life of a reactant is the time it takes for its concentration to fall to one-half of its original values. Do worksheet question #4: chemical reactions: a molecular view. In order for a reaction to occur, collisions between molecules must have: As temperature increases, more collisions between reactants have the energy necessary for reaction to occur: activation energy (ea) Minimum energy that reactant molecules must possess to undergo a particular chemical reaction: collision geometry. Some collision may have enough energy but not the right orientation: arrhenius behaviour. The arrhenius equation describes the relationship between the rate constant (k) and temperature (t). k=ae -ea/rt lnk = -e a /r x (1/t) + lna. A is called the frequency factor: reaction mechanisms. A reaction mechanism is a collection of one or more molecular steps that account for the way reactants become products. [ no2(g) + no2(g) no3(g) +no(g) ] + [ no3(g) + cp(g) no2(g) + co2(g) ] =
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232