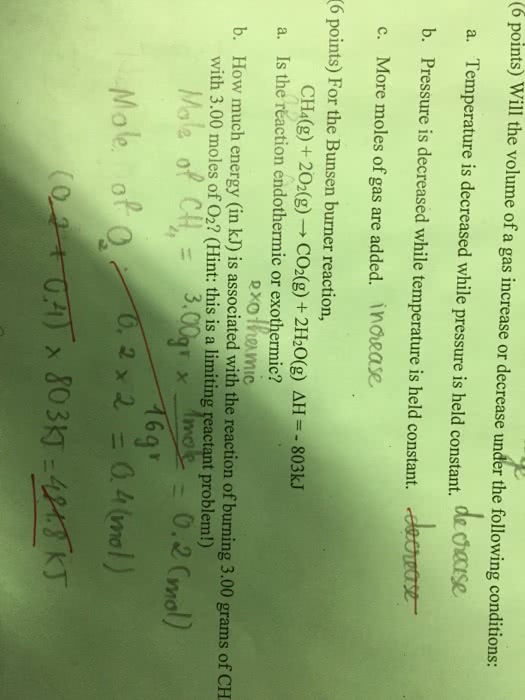CHEM 154 Lecture Notes - Lecture 17: Petri Dish, Joule, Enthalpy
88 views3 pages
Document Summary
If w = -p v, then w = 0. 10. 1 kj c: +10. 1 kj, none of the above. Answer: d: a certain chemical reaction absorbs 10. 1kj of heat under . Constant volume conditions and absorbs 8. 4 kj of heat under . Conditions. what is: the sublimation of dry ice is described by the following reaction: (we"re doing this at constant pressure) q=mc t . Enthalpy is the heat at a constant pressure. Enthalpy (h) =heat released/absorbed during chemical reaction enthalpy has units of joules (j) or kilojoules(kj). Heat is required to overcome imfs, or. Heat is released upon the formation of imf"s. H phase change n= number of moles of substance. H is an extensive property. therefore it scales with size/quantity/extent. How can we measure the enthalpy of reactions that are very slow? (or don"t happen at all?) H overall = h1 + h2: reversing reaction inverts sign of h.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232