CHEM 123 Lecture Notes - Lecture 4: Heat Capacity, Enthalpy, Military Cross

48
CHEM 123 Full Course Notes
Verified Note
48 documents
Document Summary
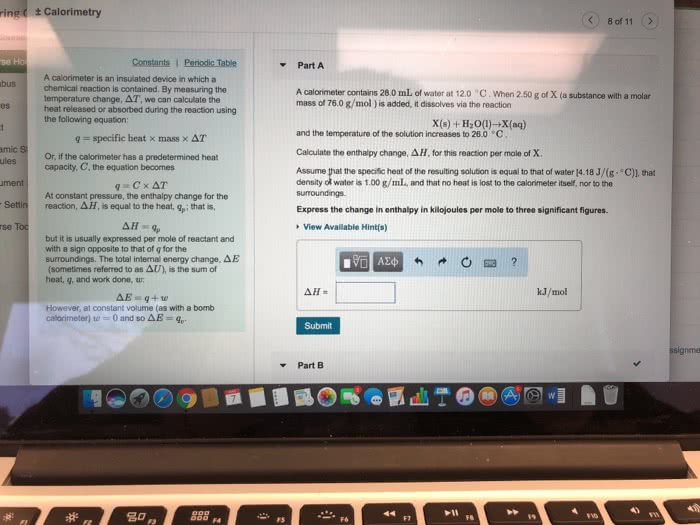
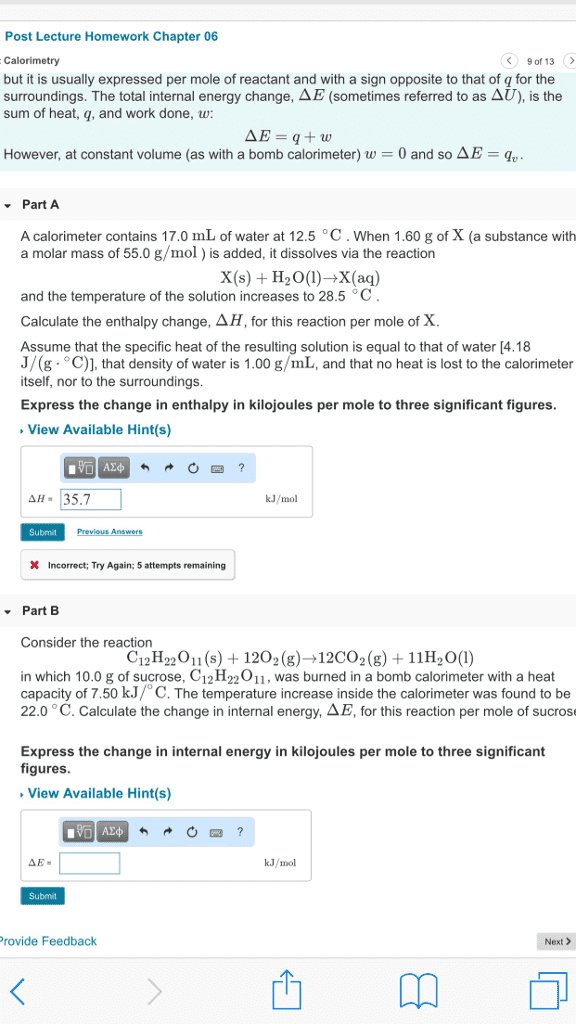
Measure heat: measuring heat and work, heat capacity, enthalpy. Change in e for that process = q = heat flow. Q describes all paths from same initial and final states value for change in e. Calorimeter definition: measures amount of heat released/absorbed in chemical reactions. Definition: amount of heat required to raise temperature of an object by one degree. Energy is added to surroundings/absorbe d from surroundings. Celcius, what is the energy change associated with the combustion of a. Intensive state variable: molar heat capacity, q = nc*change in t, specific heat capacity (mass, q = mc*change in t. Given csp (liquid h20) = 1 cal/g*degrees c. Given l change to mass via h20 density (1g/ml): mh20 = 10l* (1000ml/l)* (1g/1ml) Q = (10,000g)(1 cal/g*degrees c)*(28 degrees c) = 280,000 cal. 1 food cal = 1000 cal 280 cal. Change in e = q + w. Beginning of measuring heat at constant pressure (coffee cup.