CHEM 1050 Lecture Notes - Lecture 3: Bond-Dissociation Energy, Spontaneous Process, Covalent Bond
Document Summary
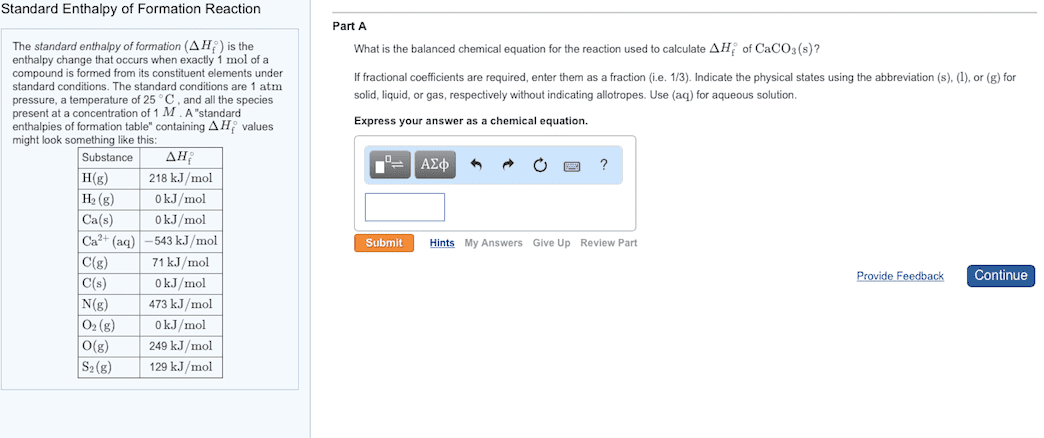
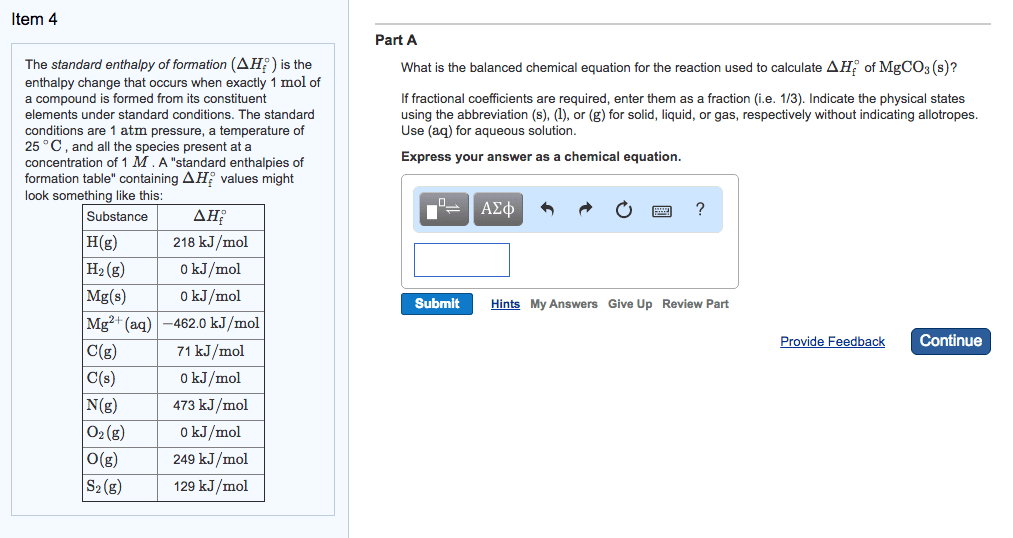
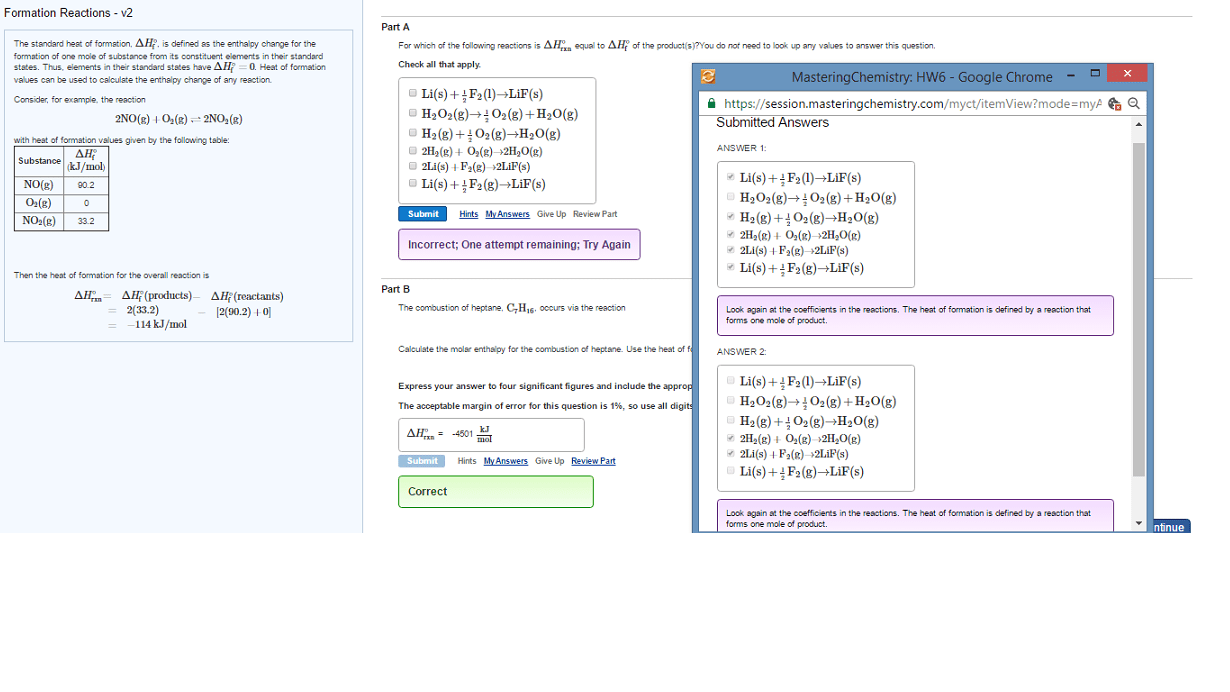
We need access to tables of delta h values. Standard enthalpy of formation (delta h knot) values are the enthalpy changes seen when we form one mole of substance from its elements in their standard states at 1. 0 x 10 ^ 5 pa (1 atm) of pressure. Delta h formation for pure elements in the standard state at a chosen temperature is equal to 0. Vap (vaporization reaction): liquid goes to a gas. To deduce the delta h formation values for all ions in an aqueous solution, we need a method to separate the contribution of the ions. Our solution, we define the following: delta h formation (h+, aq) = 0. The energy required to break one mole of bond in the gas phase only it requires energy to break the bonds. Be is the measure of the strength of a covalent bond. Note: bond dissociation enthalpy calculations are for gasses only!!!