CHEM261 Lecture Notes - Lecture 2: Formal Charge, Nitrite, A Reminder

1
CHEM261 Full Course Notes
Verified Note
1 document
Document Summary
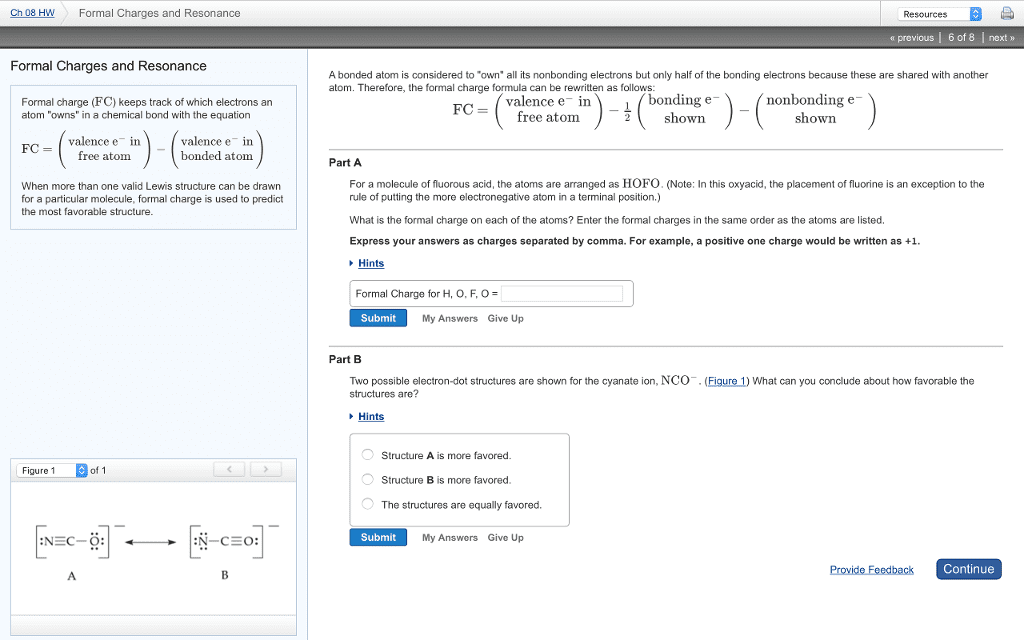
(sum of) of formal charges on all atoms in a molecule = overall charge on molecule. Subtract of the number of shared outer shell electrons. X 2 = -1 (1/2 of shared electrons) Overall charge on the nitrite anion is = -1: methyl anion. Overall charge on the methyl anion is = -1. X 6 = -3 (1/2 of shared electrons) Overall charge on the methyl anion is = 0: methyl cation (sp2 hybridized carbon, planer shape) can be reactive intermediate in principle. Overall charge on the methyl anion is = +1. Nb:the above compound can also be represented in the following forms, resulting from the free rotation of single bonds (sigma). Resonance structures: different drawings of the same molecule made by moving electrons but not atoms. Move the electrons, keeping the position of the atoms same. Maintain inert gas configuration around each atom. Double headed arrow ( ) is used indicate resonance forms.