CHEM164 Lecture Notes - Lecture 8: Stereocenter, Lone Pair, Absolute Configuration
Document Summary
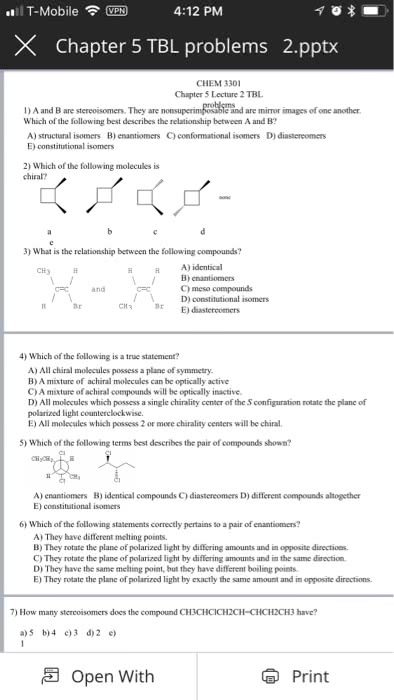
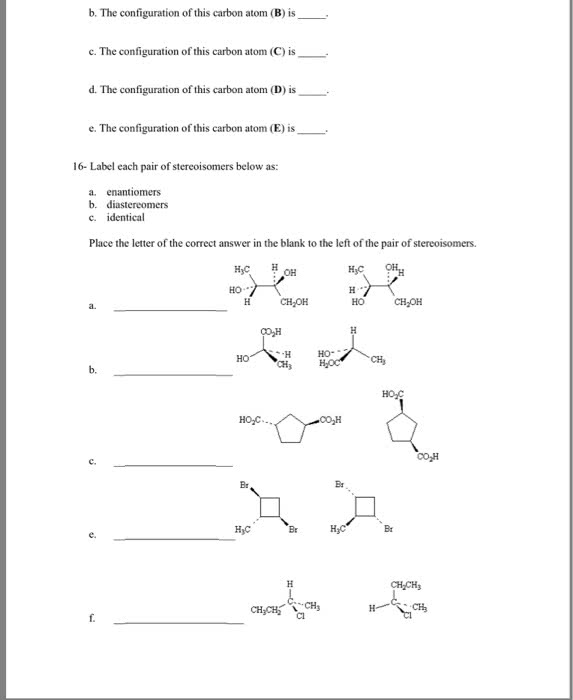
Isomers different molecules, same set of atoms. Constitutional isomers same chemical formula, atoms connected in different ways. Stereoisomers same atoms connected in the same order but are different in their 3-d arrangement of atoms. Configuration - permanent 3-d arrangement of the bonds that connect the atoms. Enantiomers molecules that are non-superposable mirror images. Superposbale - a term that describes the ability to precisely overlap one object over another. Only identical objects are superposable, everything else is non- superposable. Chirality ability of objects to exist as non-superposbale mirror images of each other. Achiral objects that are superposbale on their mirror images. Most typically a tetrahedral carbon with 4 different groups attached. Chirality centers atoms connected to four different groups. Also known as asymmetric centers, stereo genic centers, or stereocemters. A molecule with 1 chirality center has an enantiomer. The enantiomer can be produced by swapping positions of any 2 groups at the chirality center.