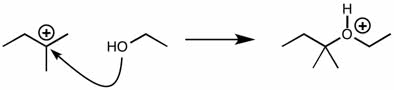
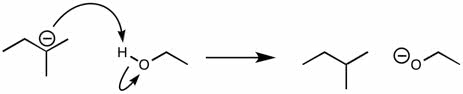
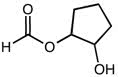
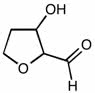
CHEM101 Lecture Notes - Lecture 10: Earl, Aufbau Principle, Paramagnetism

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
Note: 4s is more stable than 3d: electron configuration, rules for assigning electrons to orbitals. Electrons are assigned from the lowest available orbitals (aufbau principle: electronic configuration of ions. Formation of cations: cations of the main group elements are formed by losing electrons until a noble group gas election configuration is achieved eg. na na+ + e- [he] 2s2 2p6 3s1 {[he] 2s2 2p6} > [ne] Formation of anions: anions of main group elements are formed by gaining electrons until a noble group gas electron configuration is achieved eg. s + 2e- s2- Transition metal ions: fe ------------> fe2+ + 2e- [ar] : fe ------------> fe3+ + 3e, for transition metals, 4s is removed before 3d, magnetic properties of elements. Paramagnetic materials are attracted by a magnet (eg. fe) Diamagnetic materials are not attracted by magnets (eg. mg) Elements or compounds with unpaired electrons are. Elements or compounds with paired electrons and are.