CHEM101 Lecture Notes - Lecture 1: Beryllium Chloride, Lone Pair, Lewis Structure

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
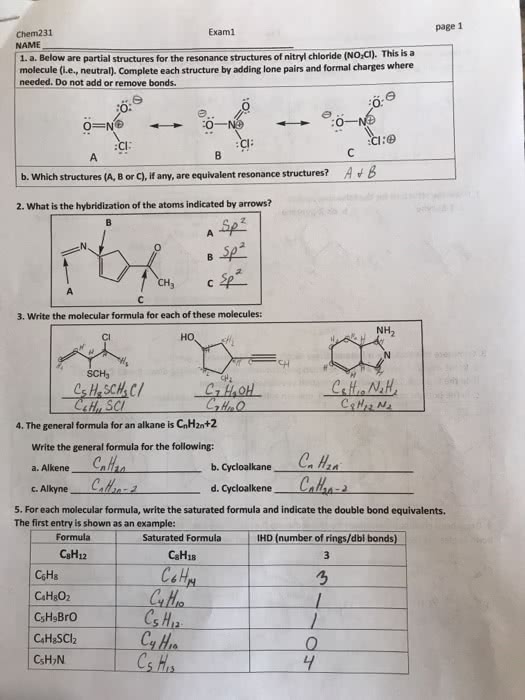
Resonance: molecule can be represented by more than one lewis structure. A double headed arrow ( , not the same as equilibrium arrows) shows resonance structures. Mostly occur in presence of both e- lone pairs and . X = y - z x - y = z. X = y = z x - y z x y - z. Never move relative positions of the atoms for each res. Structure - change only the multiple bond position. **not real - does not depict real structure of molecule. All the bonds are constantly moving, creating the resonance structures. Resonance structures are drawn since e- are not localized; they are delocalized. Single bonds are longer than double bonds - the co3(2-) structure"s bonds are equally 134 pm (in between single and double) Bond order - can be fractional; sum of each bond"s value divided by the amount of bonds.