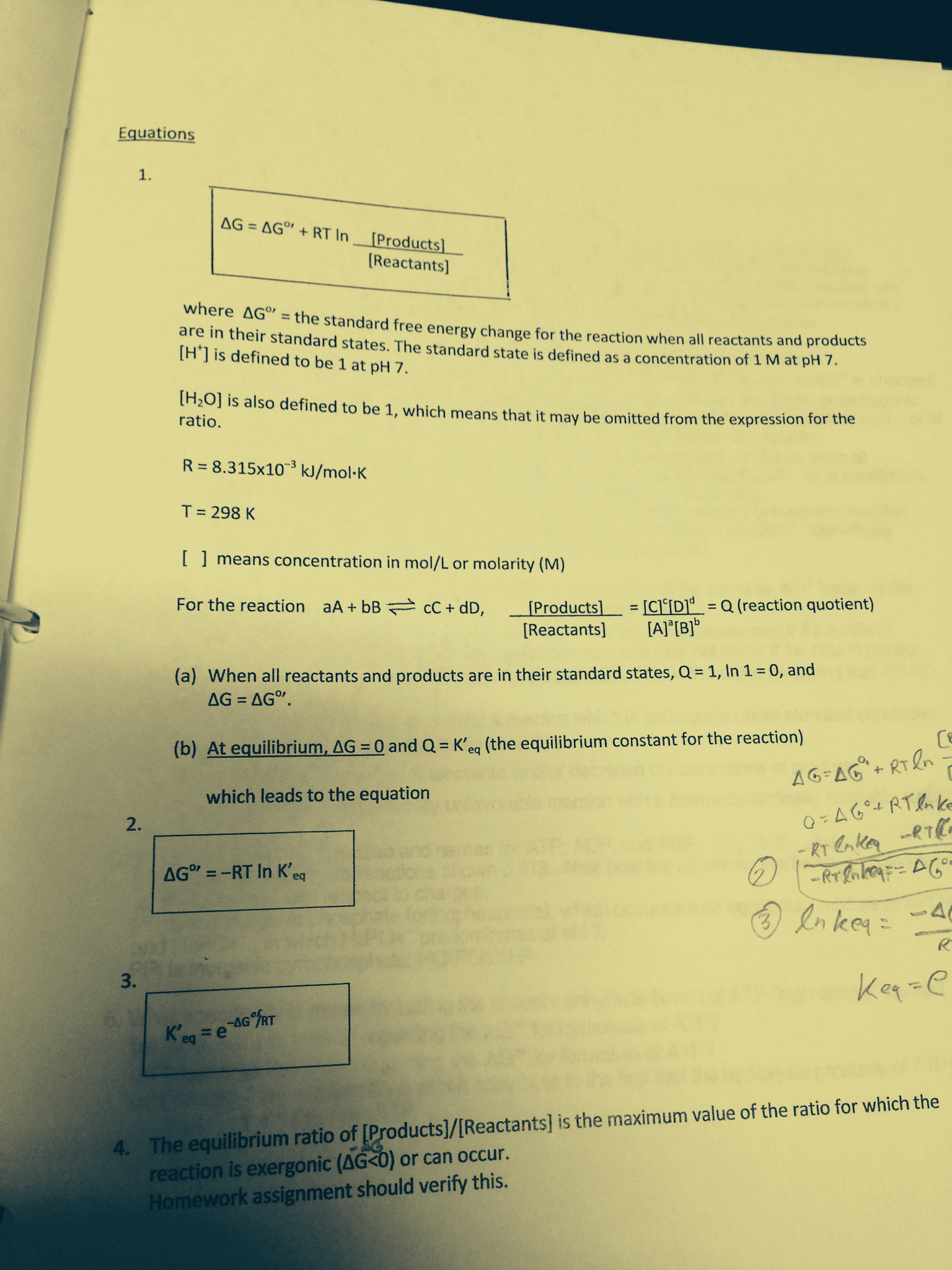BCH 261 Lecture Notes - Lecture 2: Protein Kinase, Direct Energy, Firefly Luciferin

Page 1 of 11
Ryerson University – Winter 2018
BCH 261-011&071 Biochemistry - January 31st, 2018
BCH 261-011&071 Biochemistry - January 31st, 2018
Energy in Biological Systems
Gibbs free energy, ΔG and ΔG0
• Difference between the two: ΔG0 vs ΔG
o ΔG0
○ Free energy change under standard state conditions
○ 1 M solute, Pressure: 1 bar, Temperature: 298 K
o ΔG
○ Free energy change currently happening in the reaction under current conditions
ΔG = ΔG0 + RTlnQ
Q = reaction quotient
R = gas constant
T = temperature
• Calculating free energy
Actual free energy changes depends on reactant and product concentrations
○ At equilibrium ΔG = 0
○ Substituting ΔG = 0 into the equation
= relationship between standard state free energy ΔG°& the equilibrium constant Keq
ΔG0 = -RTlnKeq or Keq = e-ΔG0 /RT
Constants and Units
● R (Gas Constant) = 8.314 J*mol-1*K-1 (or J/mol*K)
● K (Boltzmann Constant) = 1.381 * 10-23 J*K-1 (or J/K)
● 1 cal = 4.84 J
● ΔG and ΔH are J/mol, ΔS = J/mol*K
● Make sure to be familiar with these units and converting them

Page 2 of 11
Ryerson University – Winter 2018
BCH 261-011&071 Biochemistry - January 31st, 2018
Thermodynamic processes
Most processes in the cell are thermodynamically unfavourable
- Ex: one molecule of RNA is made from millions of nucleosides
- Ex: Thousands of amino acids make up one molecule of a protein
Favourable processes (Processes that have a negative ΔG)
- Ex: Cells use sugar/fat which have high energy content. The sugar and fat are used to power
reactions that aren’t favourable such as making RNA and proteins, or moving muscle
ATP is the energetic currency in cells
● Cells obtain free energy from either chemical oxidation or sunlight
● Cells use energy to synthesize ATP and that ATP is spent on unfavourable processes
● ATP referred to as a high energy bonding which is a misnomer. ATP is actually a high energy
phosphate compound, the high energy is not in the breaking of the bond.
Ø figure shows the phosphoanhydride bonds within ATP and ADP

Page 3 of 11
Ryerson University – Winter 2018
BCH 261-011&071 Biochemistry - January 31st, 2018
Chemical basis for large and negative free energy with ATP hydrolysis
● The charge separation from hydrolysis relieves electrostatic repulsion among the 4 negative
charges on ATP
● The product- inorganic phosphate (Pi) is stabilized by formation of a resonance hybrid
○ each of the 4 phosphate oxygen bonds and the H+ ion is not permanently associated
with any one of the hydrogens
● Greater degree of solvation on the products Pi and ADP relative to ATP which further
stabilizes the products relative to the reactants
○ Ionized immediately because the concentration of the direct products of ATP
hydrolysis are far below the concentration at equilibrium
○ Actual free energy of hydrolysis (ΔG) of ATP in living cells is very different because
■ The cellular concentration of ATP, ADP, and Pi are not identical
■ ATP, ADP, and Pi are much lower than 1.0 in standard condition
■ Mg2+ in the cytosol binds to ATP and ADP
Document Summary
Gibbs free energy, g and g0: difference between the two: g0 vs g. Free energy change under standard state conditions. 1 m solute, pressure: 1 bar, temperature: 298 k. Free energy change currently happening in the reaction under current conditions. Actual free energy changes depends on reactant and product concentrations. Substituting g = 0 into the equation. = relationship between standard state free energy g & the equilibrium constant keq. G0 = -rtlnkeq or keq = e- g0 /rt. R (gas constant) = 8. 314 j*mol-1*k-1 (or j/mol*k) K (boltzmann constant) = 1. 381 * 10-23 j*k-1 (or j/k) G and h are j/mol, s = j/mol*k. Make sure to be familiar with these units and converting them. Most processes in the cell are thermodynamically unfavourable. Ex: one molecule of rna is made from millions of nucleosides. Ex: thousands of amino acids make up one molecule of a protein. Favourable processes (processes that have a negative g)