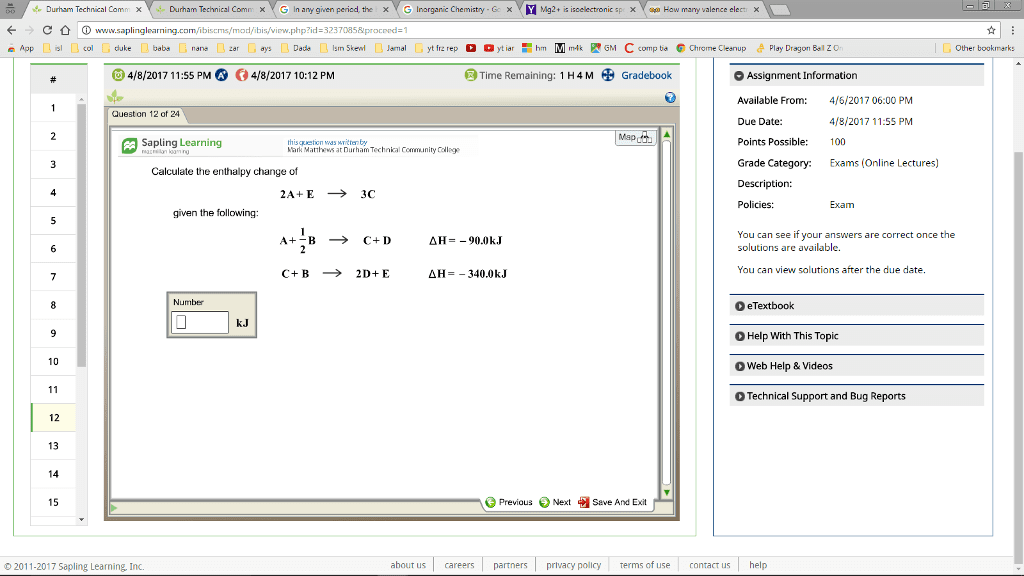
Heat of Neutralization Lab Partner Date Reaction 1 Reaction 2 Reaction 3 1. Initial temperature 2. Final temperature 3, The change in temperature, AT +2.o 2 o c 30 c 4. Heat, Q, absorbed by the solution mass of soln. x AT x 1 Note: Mass m sum of masses of solids G.15 4.2 and liquids used. EG geax 5. The amount of heat absorbed by Gut cal the cup (neglect this quantity) neglect neglect 6, TOTAL amount of heat. Q absorbed 303 Moca. Guogsal y10.ou cal 7. The number of moles of NaoH o out out used in each reaction For solid: 1/MM NaoH o.ou mol For solution: L. NaoH xMi 8. The heat of reaction per mole ofNaoH at to AH. Include sign. (Line 6/Line 7) Questions 1, write the net ionic equations for reactions 1.2, and 3 equations for reactions 2 and 3 and make a statement concerning the significance of 4H2. that is. what has happened to produce the energy change. Also explain the mean- ing of 4H3. 3. How does 4H2 compare with the sum of AH1 AH3. Explain. 4. Calculate the percent difference between AH2 and the SUM of AH1 AH3, using AH2 as the reference. Difference x 100% difference