CHEM 2OA3 Lecture Notes - Lecture 1: Cyclohexane, Heptane, Chloromethane
Document Summary
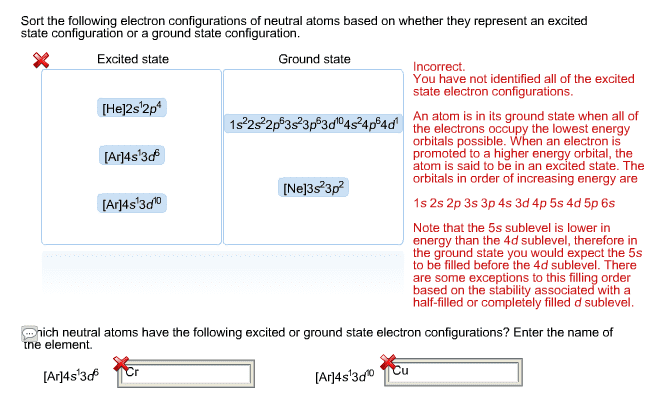
14 may 2015: 1-6 of this tutorial is based on the first two lectures and chapter 1 of. Q. 7 is based on podcast 2, while q. 8 is based on podcast 1. Give the electronic configurations of the resulting excited states of these atoms, by filling in the correct numbers of electrons on the template below: Indicate the polarity, if any, of the following bonds to carbon, using the +/ - convention: Nitromethane ( nitro , ch3no2) is used as a fuel in drag car racing. (a) Draw a lewis dot structure for nitromethane, showing all the individual atoms, bonds, charges and lone pairs of electrons. Nitromethane has two equivalent resonance forms - draw them. (b) (c) methyl nitrite is a structural isomer of nitromethane. Draw a structure for methyl nitrite, showing all bonds, lone pairs, etc For molecule 1, in which the carbon atoms are numbered according to the iupac convention, answer the questions below.