CHEM 1A03 Lecture Notes - Lecture 1: Ionic Compound, Intramolecular Force, Ionic Bonding

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
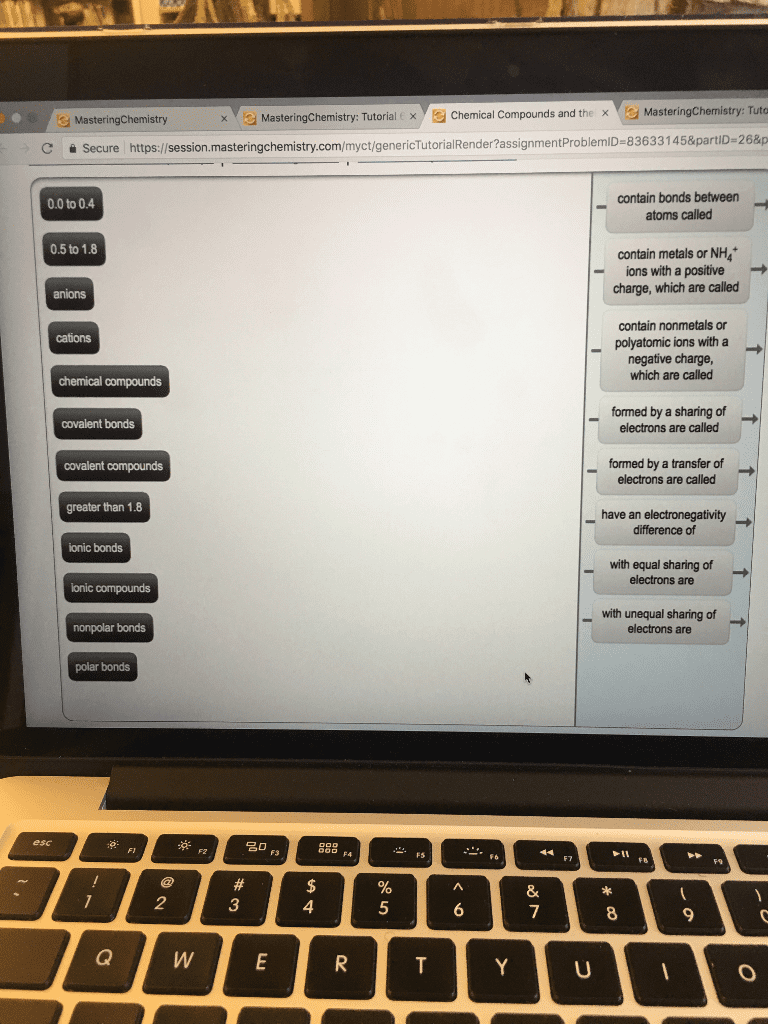
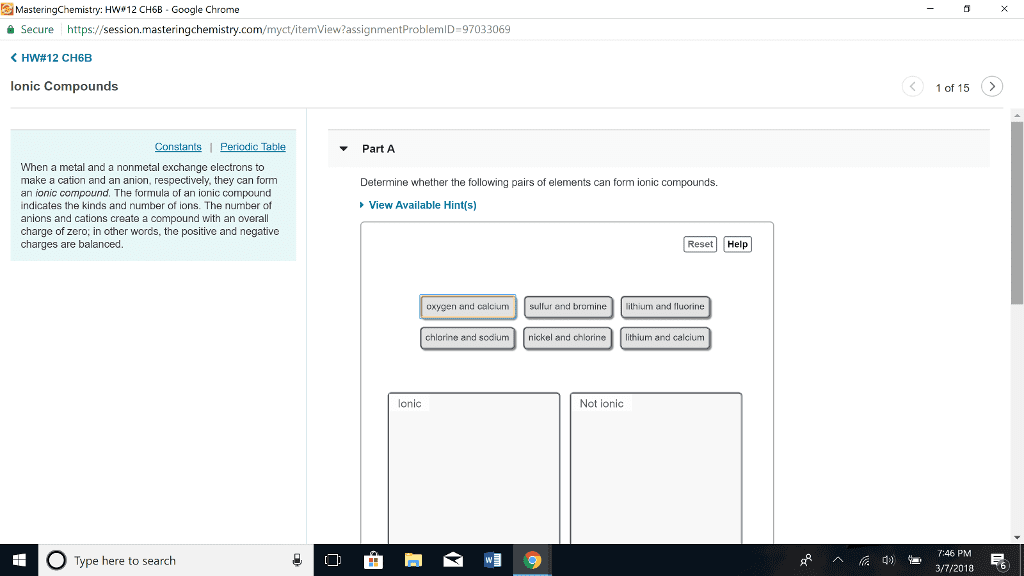
Ionic compounds are pure substances composed of ions and formed by the transfer of electrons from a metal and a nonmetal. Ionic bonds are a type of chemical bond created due to the electrostatic attraction between positive and negative ions. Octet rule states that representative elements want a stable octet consisting of a full shell of eight electrons in their outer energy level. Metallic elements lose electrons (positive ions or cations) Nonmetallic elements gain electrons (negative ions of anions) Molecular compounds are pure substances composed of two or more nonmetals. Covalent bonds are a type of chemical bond created by an attractive force between two atoms when they share electrons. Nonmetallic elements want to gain electrons and are involved with covalent bonding. The simplest whole number ratio of ions in an ionic compound is called the formula unit. ions actually form a crystal lattice a regular, ordered arrangement of ions. Electical conductivity in solid state in liquid state.