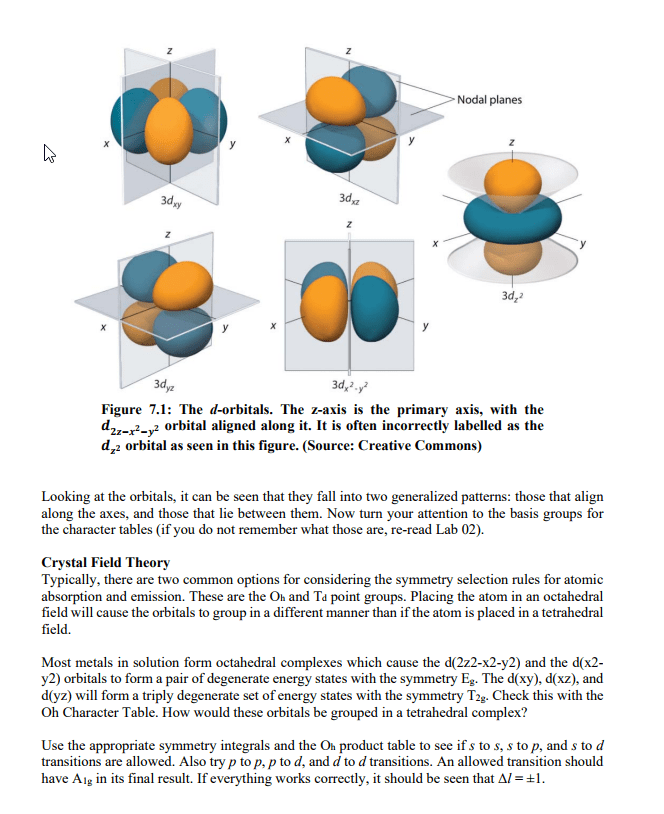
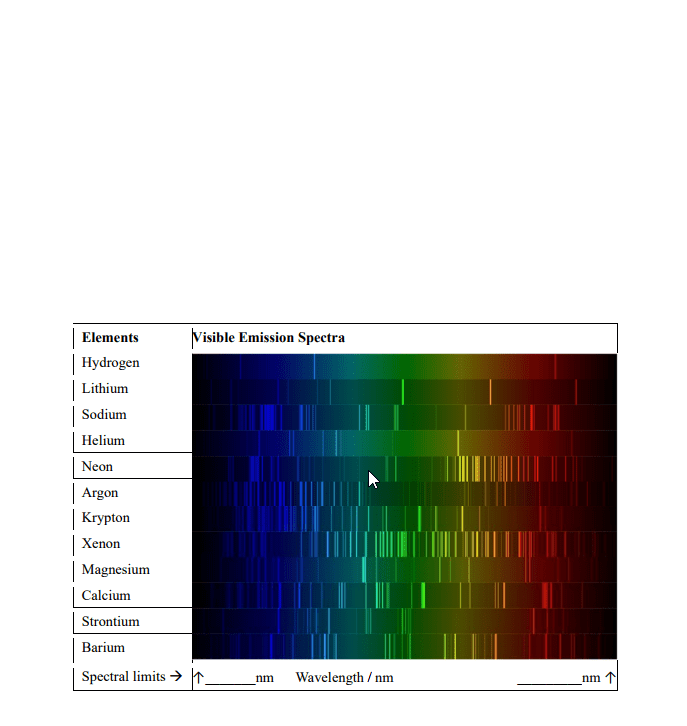
CHEM 1A03 Lecture Notes - Lecture 3: Threshold Energy, Rydberg Constant, Balmer Series

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
All light is made up of electromagnetic waves. Composed of an electrical component and a perpendicular magnetic component travelling in the same direction. Wavelength ( ) = distance between peaks or troughs. Frequency ( v ) = wavelengths / time. Speed of light ( c ) = 2. 9979 x 108. Properties: energy frequency, energy 1/ . Each element has a unique line spectrum. Contains a nucleus, composed of neutrons and protons. Nucleus is relatively small but contains almost the entire mass. Electrons surround the nucleus, acting as particles and waves. Energy is discontinuous and comes in quanta. Each quantum of energy is a photon. Energy of a photon = frequency x planck"s constant. E = h v ; h = 6. 626 x 10-34j/s. No circuit, plates separated by a vacuum. Only way to complete the circuit is to cause an electron to jump from one plate to another.