CHEM 222 Lecture Notes - Lecture 8: Markovnikov'S Rule, Syn And Anti Addition, Halohydrin
Document Summary
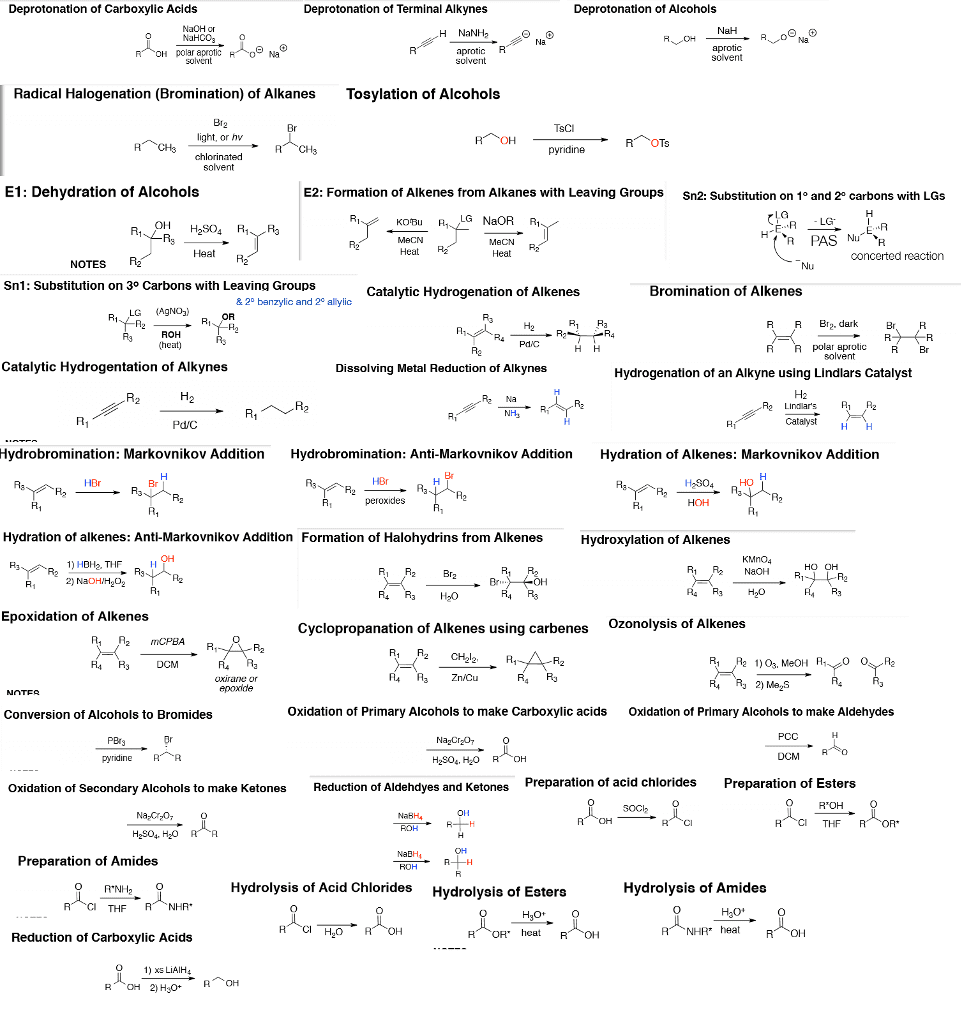
If sn1 or e1 on the midterm, and ask for major product, could write both and say present as racemic mixture. In an alkene, the functional group is the pi bond: addition of strong acids (hx), of weak acids (h2o), with cyclic ts (making alcohols), addition of (xx) Eliminations: the acid in this case acts as a catalyst, gets regenerated, this is a reversible reaction. *it might not be h-x might be i-br or other halogens, just take the most electronegative as x and least as h. Epoxide formation: used for glue, insects (moth) Reactions of epoxide: occurs since results in release of the ring strain, wouldn"t otherwise, cis remains cis when make epoxide, backside attack, ohs added in anti fashion. Oxymercuration-demercuration: markinov addition, non-stereoselective (syn & anti, no rearrangement! Oxidation alkenes: formal oxidation state: not the charge of the carbon, only there to keep track of the oxidation/reduction in reactions.