CHEM 212 Lecture Notes - Lecture 6: Steric Effects, Weak Base, Electronegativity
99 views7 pages
17 Jun 2015
School
Department
Course
Professor
Document Summary
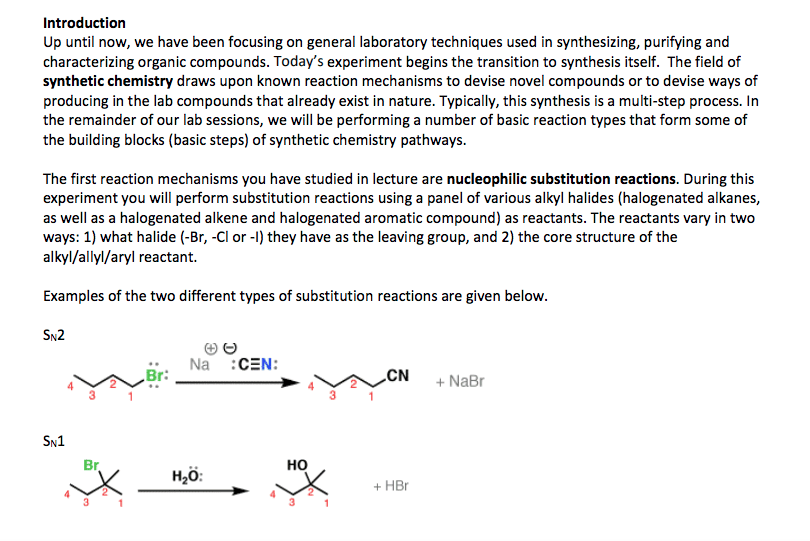
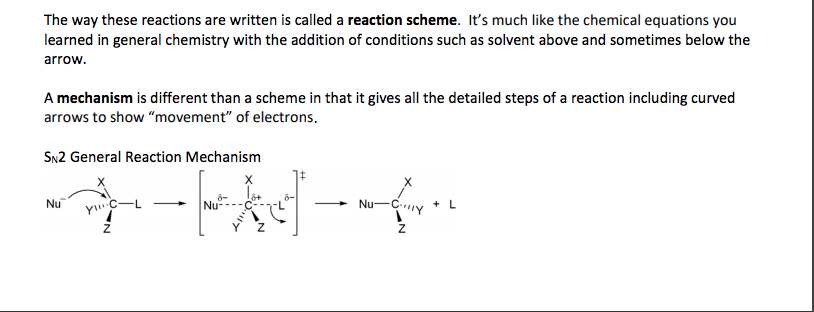
Chem 212: introductory organic chemistry 1 lecture 6: ionic reaction (part 1) Electrophile: carbocation or + end of a polar c x bond. Nucleophile: attacking lewis base (can be neutral or anionic) Leaving group: displaced halide ( end of a polar c x bond) Substitution reactions = nucleophile attacks electrophile (with displacement of leaving group. Rate of reaction: the change in concentration of the reactants or products per unit time. Determining order: measure rate at varying initial concentrations of a and/or b and determine the effect on the rate. Rate = k[a]s[b]t; order = s + t. Doubling [a] doubles rate (s = 1) Doubling [b] no effect on rate (t = 0) Doubling [b] doubles rate (t = 1) Two separate steps: bond breaking to lg, then bond making to nu. S = substitution; n = nucleophilic; 1 = unimolecular. In this reaction, the first step is the slowest, and therefore, the rate determining step.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232