CHEM 120 Lecture Notes - Lecture 6: Chemical Kinetics, Sodium Chloride, Rate Equation

26
CHEM 120 Full Course Notes
Verified Note
26 documents
Document Summary
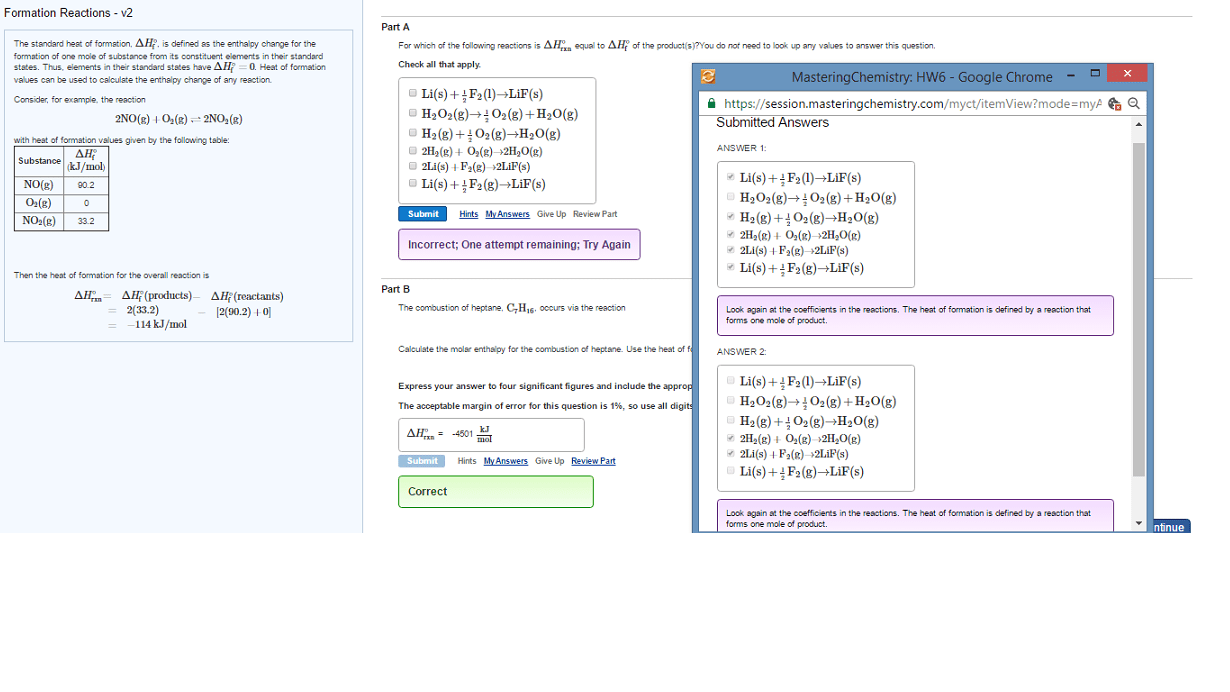
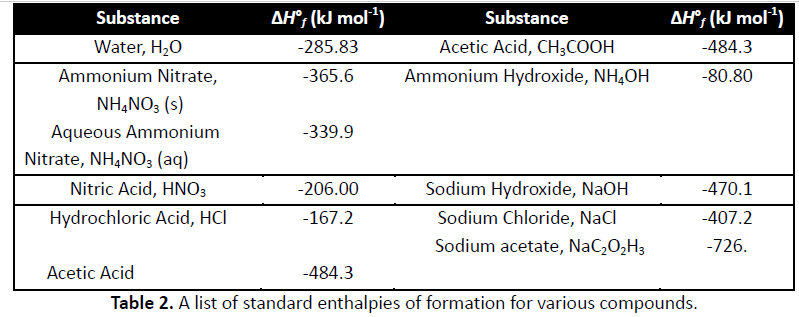
Jan 24 2019: lecture 6: thermo cont"d and kinetics. The enthalpy change that occurs in the formation of one mole of a substance in the standard state from the reference point of the elements in their standard states. Elements in their pure form have deltafho = 0. Reactants decompose to elements and then elements rearrange and organize into products. If we know deltafho reactants and deltafho products, then. Deltarho = sum(mdeltafho products) - sum(ndeltafho reactants) Where n and m are number of moles. Chemical kinetics measures the rate at which the concentration of a substance ( reactant or product) changes with time. In this reactions, as time elapses [hf] will increase as [h2] and [f2] decreases. Ab + bb cc + dd. If we can measure the concentration of reactants and products at different intervals of time, then we can plot the change in the concentration of reactants/products as a function of time.