CHEM 120 Lecture Notes - Lecture 23: Electroplating, Ion, Radioactive Decay

Nerst Equation: non-standard/standard conditions are related;
-
-
When [products]/[reactants] = 1 that is standard conditions; still have Ecell
- Q < 1, cell does more work, favouring the reactants
- Q = K, cell does no work; E
Electrolysis: electrical energy is used to drive a non-spontaneous reaction
- e is drawn from anode, charge labels are opposite of Galvanic cells
- cathode (reduced), anion (oxidized)
Molten Salt Mixtures: to figure out which metal is the cathode, the highest IE is the one who
will be reduced at the cathode (Mg beats Na) & lowest EN atom will be oxidized (Br beats
Cl) at the anode it will release 2e
o cannot use E because it only describes 1M aq. species, just given as
E without salt
Pure Water: anode = formation of O2|cathode = formation of H2|electrolyte = dilute aq. salt
solution
Aqueous Salt Mixtures: most negative Ehalf-cell will be oxidized at the anode & most
positive Ehalf-cell will be reduced at the cathode
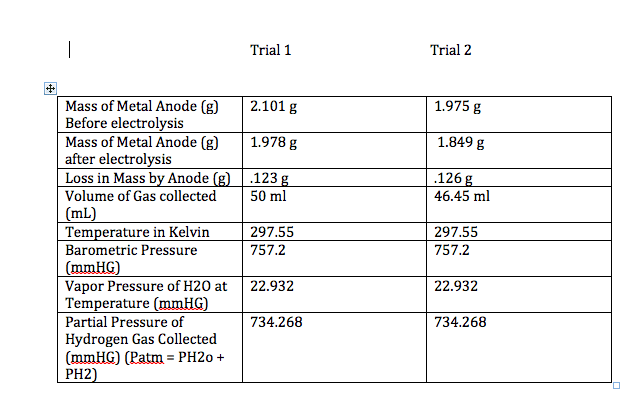
Electroplating: process of depositing 1 metal on another
Faraday’s Law: amount of substance produced at each electrode is proportional to the
quantity o charge flowing through the cell
I = Q/t and n = Q/F
1 mol e = F
NUCLEAR CHEMISTRY
Positron: positive electron | α- particle: He atom
find more resources at oneclass.com
find more resources at oneclass.com

26
CHEM 120 Full Course Notes
Verified Note
26 documents
Document Summary
When [products]/[reactants] = 1 that is standard conditions; still have e cell. Q < 1, cell does more work, favouring the reactants. Q = k, cell does no work; e (cid:1855)(cid:1857)=(cid:1866) Electrolysis: electrical energy is used to drive a non-spontaneous reaction. E is drawn from anode, charge labels are opposite of galvanic cells cathode (reduced), anion (oxidized) Molten salt mixtures: to figure out which metal is the cathode, the highest ie is the one who will be reduced at the cathode (mg beats na) & lowest en atom will be oxidized (br beats. Cl) at the anode it will release 2e: cannot use e (cid:1858) (cid:1855)(cid:1857) because it only describes 1m aq. species, just given as. Pure water: anode = formation of o2|cathode = formation of h2|electrolyte = dilute aq. salt solution. Aqueous salt mixtures: most negative ehalf-cell will be oxidized at the anode & most positive ehalf-cell will be reduced at the cathode.