CHEM 110 Lecture Notes - Lecture 7: Reaction Coordinate, Activation Energy, Exothermic Process

CHEM 110: P7 CHAP 21: ORGANIC REACTIONS
Mechanisms and Energy
Concerted Reaction: one-step mechanism, reactants are converted directly to
products (potential energy vs. reaction coordinate)
z activation energy
activation energy
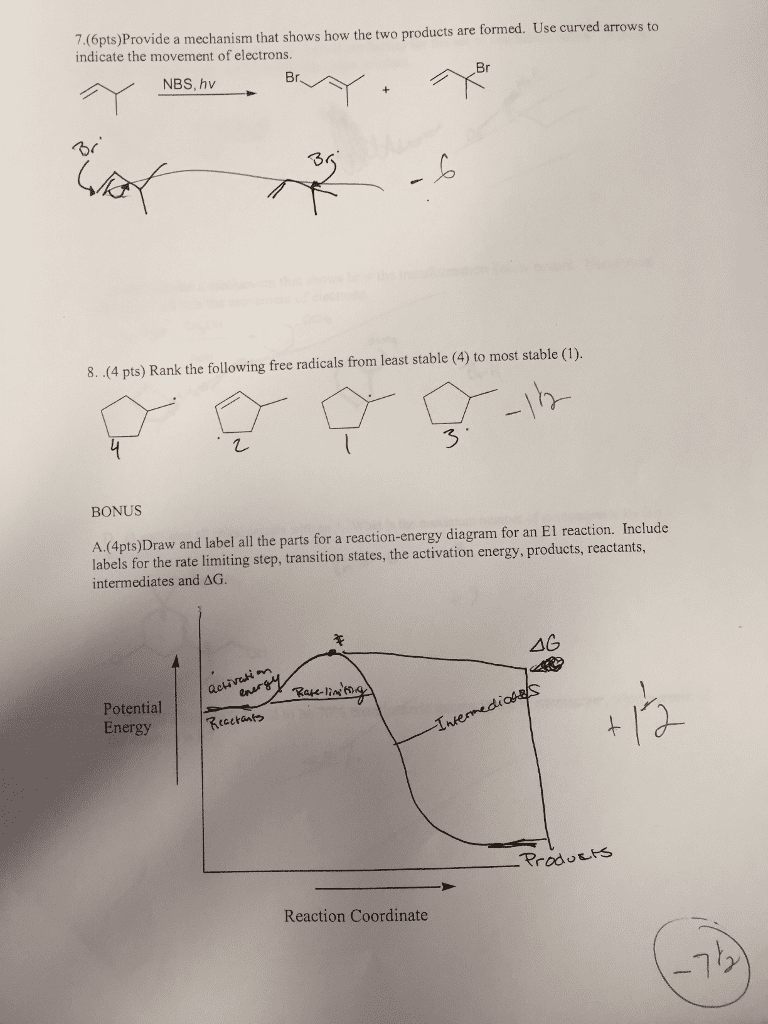
Stepwise:
Intermediate
Practice Using Curved Arrows:
Exothermic (substitution rxn)
find more resources at oneclass.com
find more resources at oneclass.com

37
CHEM 110 Full Course Notes
Verified Note
37 documents
Document Summary
Concerted reaction: one-step mechanism, reactants are converted directly to products (potential energy vs. reaction coordinate) z. Hydrogenation of oils: oils are unsaturated fats (they have double bonds) they contain alkenes, kink in the chain and can"t pack tightly, lower intermolecular forces, lower bp/mp makes liquid! Fatty acids: intermolecular forces are stronger, higher melting/boiling points. Hydrogenation the addition of hydrogen to an unsaturated compound. *slide* net result= alkene turns to an alkane. Hydrogen sticks to metal catalyst, forms a bond, becomes weak enough for so that the alkene can take the hydrogens from the metal. Take the hydrogen"s away, turns into a trans double bond. Margarine is not used by hydrogenation anymore due to production of trans fats; which are extremely indigestible (partial hydrogenation trans fats) *slide* made from individual repeat units of the same molecule. Ex: polyethylene made from individual chain reactions of ethylene. Addition polymerization: chain-growth polymerization making polymers (alkene on alkene on alkene)