ELEC 211 Lecture Notes - Lecture 15: Electric Field, Covalent Bond
Document Summary
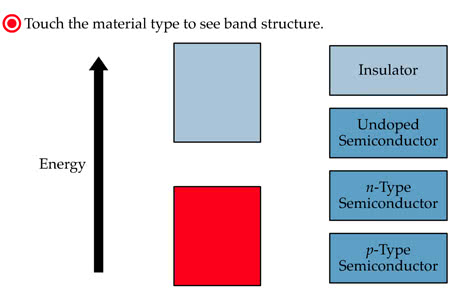
Materials can be classified by their ability to conduct electricity. This ability is related to the valence electrons. Copper is an example of an excellent conductor. It has only one electron in its valence band, which can easily escape to the conduction band, leaving behind a positive ion (the core). Like all metals, copper has many free electrons which are loosely held by the attraction of the positive metal ions. Insulators have tightly bound electrons with few electrons available for conduction. Nonmetals, such as glass, air, paper and rubber are excellent insulators and widely used in electronics. Even these materials can break down and conduct electricity if the voltage is high enough. Semiconductors are between conductors and insulators in their ability to conduct electricity. Silicon is an example of a single element semiconductor. It has four electrons in its valence band. Unlike metals, silicon forms strong covalent bonds (shared electrons) with its neighbors.