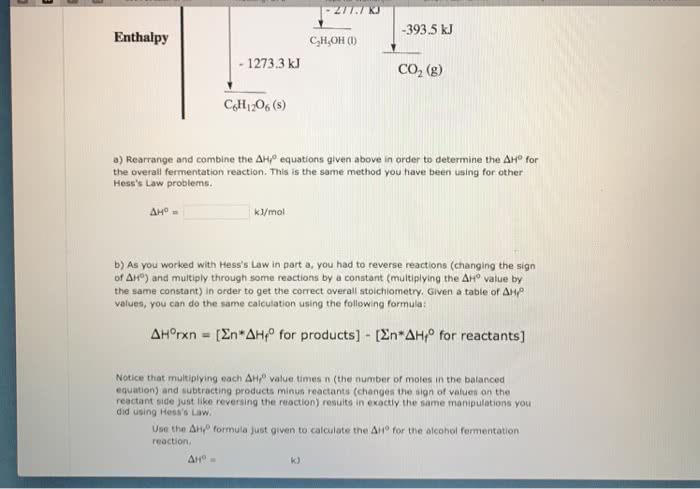
CHEM 1001 Lecture Notes - Lecture 6: Quadratic Equation, Equilibrium Constant, Stoichiometry

7
CHEM 1001 Full Course Notes
Verified Note
7 documents
Document Summary
In most reactions there is a balance between which direction a reaction goes. A one way reaction is one that is more likely to go in a certain way. However, for most reactions there is a balance between the forwards and backwards reactions, called equilibrium. With equilibrium it is likely that some reactions may have the reverse reaction happening at the exact same time. As reactants decrease to constant amount, products increase to constant amount. Dynamic equilibrium: if the rate of forward = rate of backward. This looks like nothing more is happening, the reaction has stopped . Independent of initial conditions: this concept of products to reactants ratio is the same as reaction quotient (q) but only applies to the reaction once it has reached equilibrium, q applies to any point in the reaction progression. Q = (products)^n/(reactants)^n; when not at equilibrium.