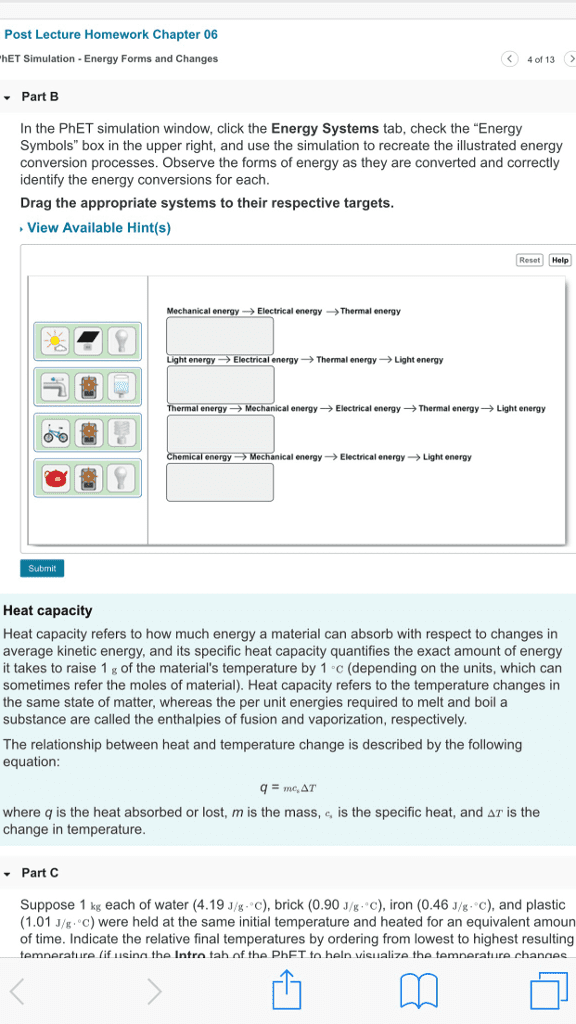
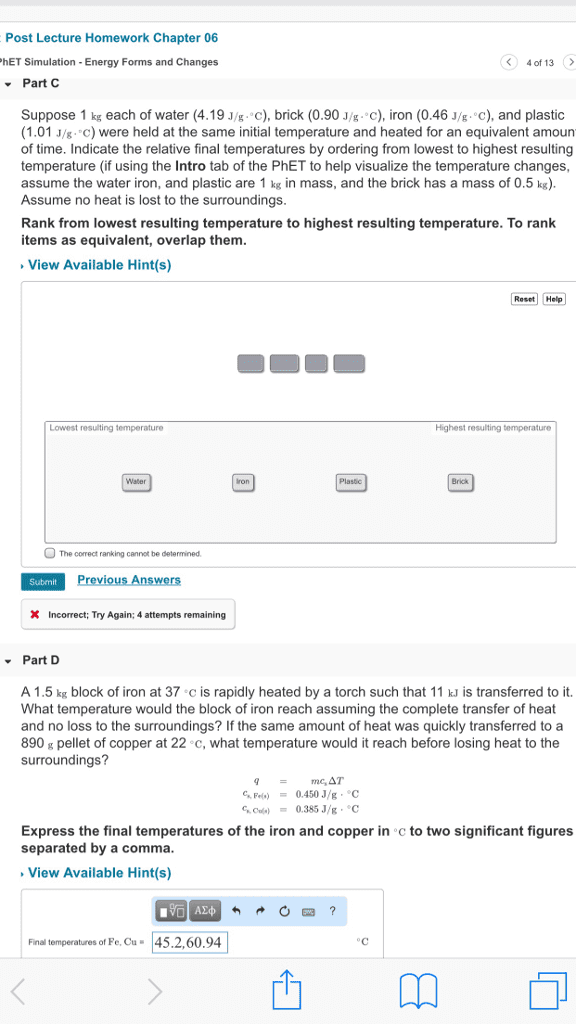
CHEM 1P00 Lecture Notes - Fall 2018 Lecture 7 - Heat capacity, Chemical substance, Magnesium
42 views1 pages
Document Summary
The amount of energy required to raise the temperature of 1g of substance by 1 degree. The specific heat capacity of material is a measure of how easy it is to health or cool something down. Example: 2. 98 calories of heat are needed to raise a temperature of 2. 00 of metal from 25. 0 degrees c to 37 degrees c. calculate the metal using the table below to determine which metal it is. = (cid:2870). (cid:2877)(cid:2873) (cid:3039) (cid:2872). (cid:2869)(cid:2876)(cid:2872) (cid:2870). (cid:2868)(cid:2868) (cid:2869)(cid:2870). (cid:2868) (cid:3039) (cid:2870). (cid:2868)(cid:2868) (cid:4666)(cid:2871)(cid:2875). (cid:2868) (cid:2870)(cid:2873). (cid:2868)(cid:4667) A chemical substance that contains only one type of atom. Has a symbol for 1 or 2 letters.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232