CHEM 1F92 Lecture Notes - Lecture 7: Ph, Alkaline Earth Metal, Beryllium
Document Summary
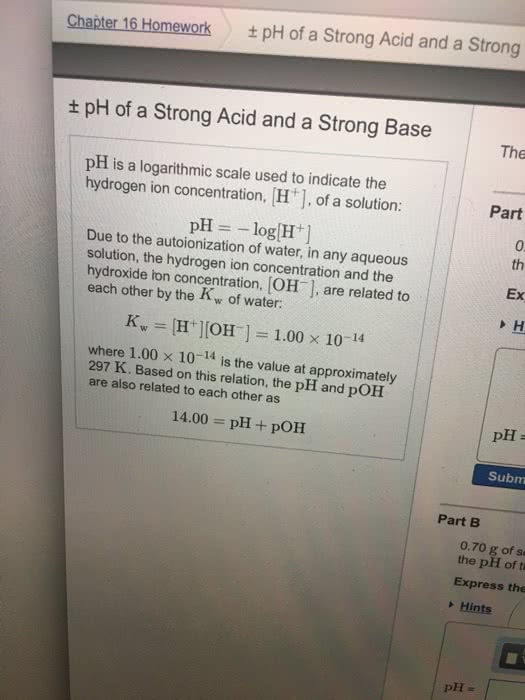
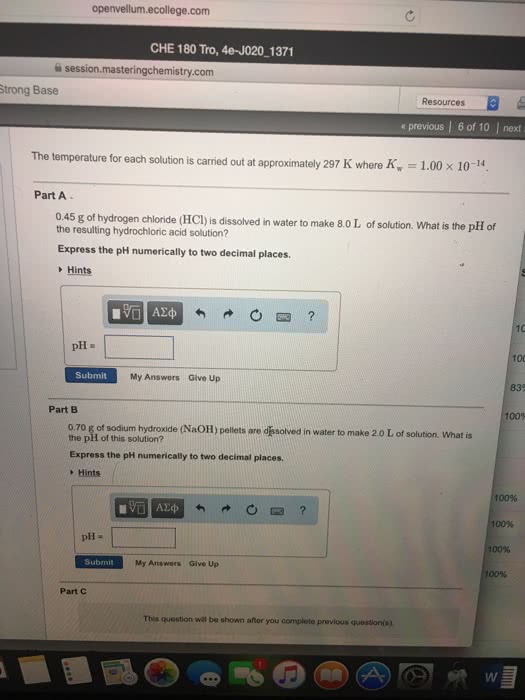
Ability to act either as an acid or a base. Acts as a base in reactions with acids. Acts as an acid in reactions with bases. Very weak electrolyte but undergoes ionization to a small extent. Kw = k[h2o] = [h+][oh-] = 10-14. In the kinetics, we look to see how the concentrations change throughout the reaction. Calculate [h+] in a solution whose hydroxide concentration is 3. 7 x 10-9 m. Method 1: ph = 14. 00 - poh poh = -log(3. 7 x 10-9) = 8. 43 ph = 14. 00 - 8. 43 = 5. 57. Strong acids dissociate completely in aqueous solutions. Hclo4, hi, hbr, hcl, h2so4 (first stage of ionization) and hno3. Hydroxides of alkali metals and of alkaline earth metals (except beryllium) Strong bases dissociate completely in aqueous solution. The poh of a strong base is 1. 88 at 25 . Calculate the concentration of the base if the base is ba(oh)2. Weak acids dissociate only partially in aqueous solution.