CHEM1011 Lecture Notes - Lecture 11: Covalent Radius, Atomic Radius, Ionic Radius
Document Summary
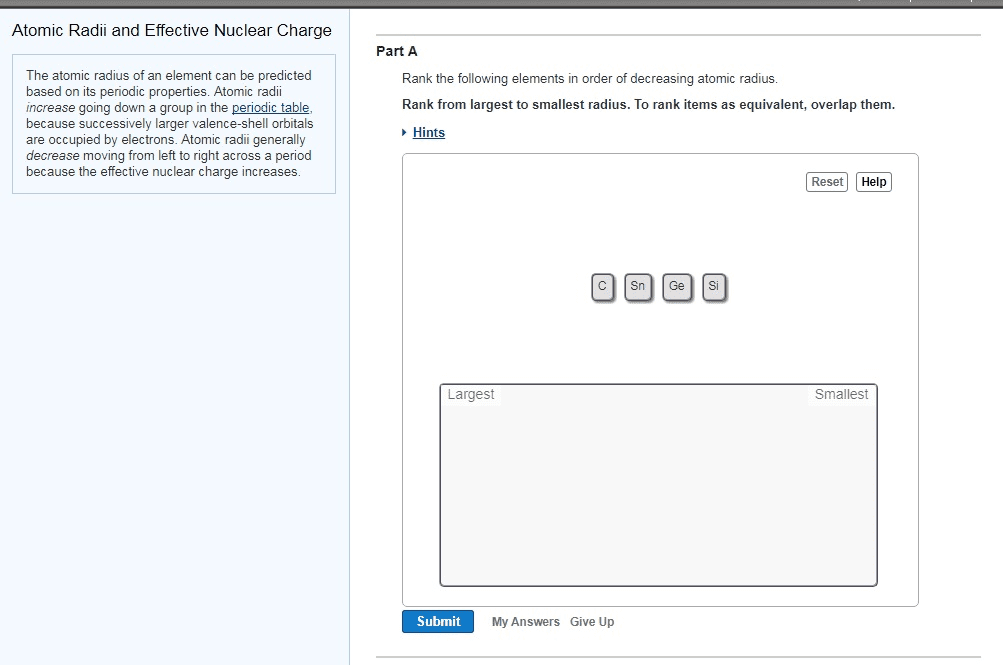
Periodicity: predicting properties of atoms, ions and bonding based on where the elements are on the periodic table. Predicting properties of atoms, ions and bonding based on where the elements are on the periodic table. In order of importance, what you need to know are: trends across a period and across a group, why these trends occur, some key anomalies, and how these may be explained. Remember electrons exist in a cloud surrounding the nucleus and as such the edge" of the atom is difficult to define. There are a number of different methods to describe the radius of an atom such as van der waals radius, ionic radius or covalent radius. For elements that exist as single atoms in the solid state (metals, noble gases) the atomic radius is defined as one half the distance between adjacent nuclei. For diatomic molecules (e. g. o2, cl2) the covalent radius is defined as half the internuclear distance.