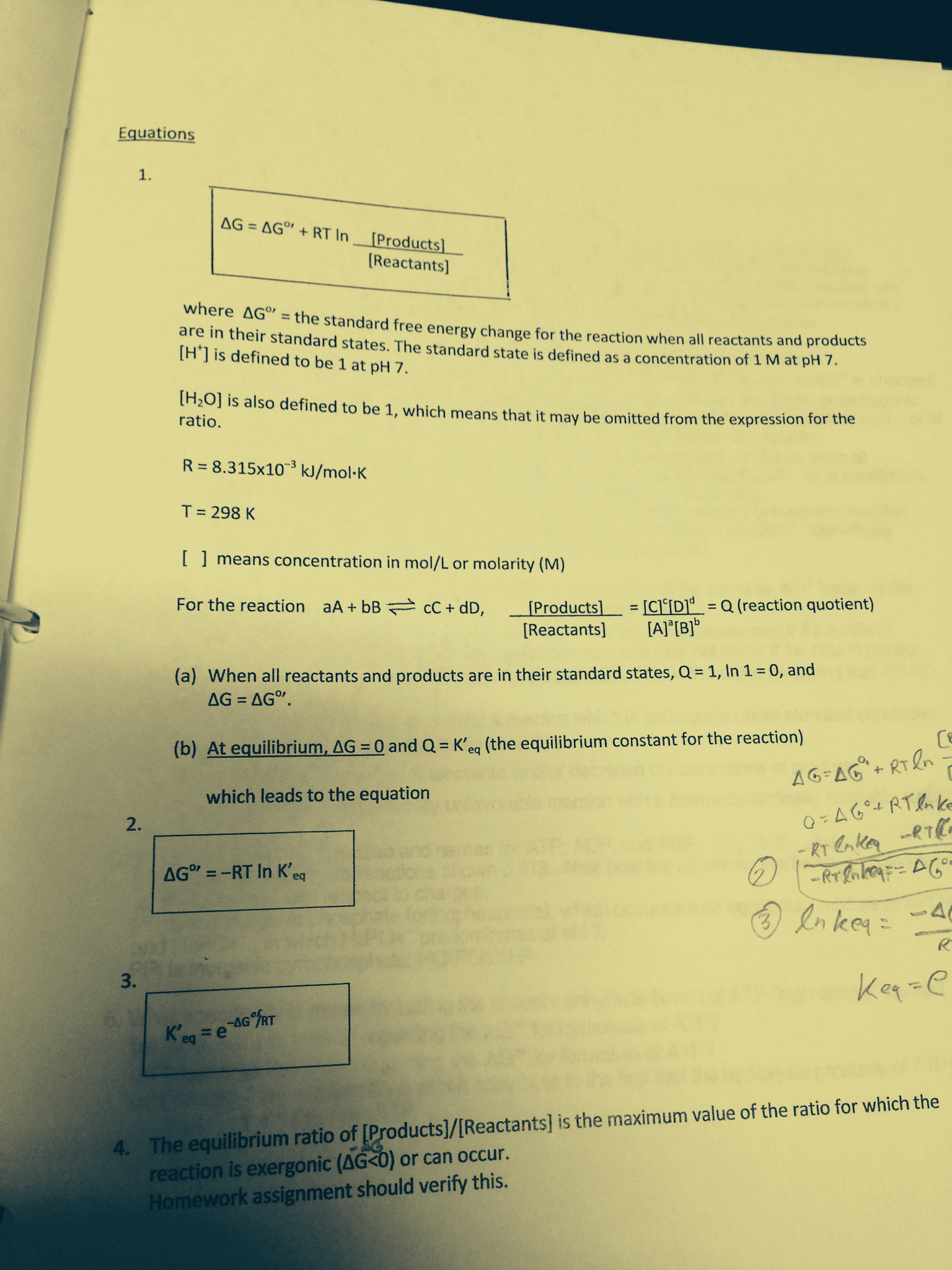CHEM10003 Lecture Notes - Lecture 24: Equilibrium Constant, Dynamic Equilibrium
Document Summary
If delta g < 0 or q < k the forward reaction proceeds. If delta g > 0 or q > k the reverse reaction proceeds. If delta g = 0 or q = k the reaction is at equilibrium. If you want to increase q, more products will need to be formed. When a change is made to a system in dynamic equilibrium, the system responds to minimise the effect of the change. If reactant is added, reaction will adjust to consume some of added reactant. If product is added, reaction will adjust to consume some of added product: for gaseous reaction, decreasing volume of container will push equilibrium in direction of fewer molecules (acts to reduce pressure) If reaction is exothermic (delta h is negative) a temperature increase will reduce value of k. If reaction is endothermic (delta h is positive) a temperature increase will increase value of k.