CHEM10003 Lecture Notes - Lecture 17: Boiling Point, Internal Energy
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
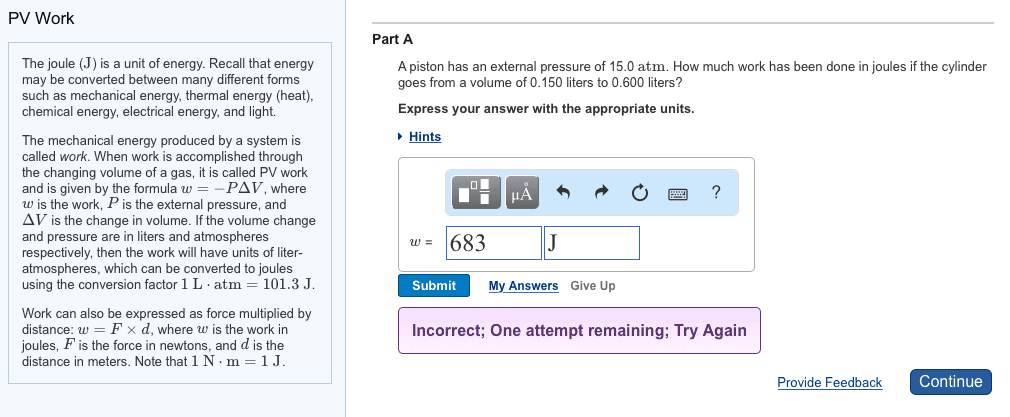
| PV Work The joule (J) is a unit of energy. Recall that energy may be converted between many different forms such as mechanical energy, thermal energy (heat), chemical energy, electrical energy, and light.The mechanical energy produced by a system is called work. When work is accomplished through the changing volume of a gas, it is called PV work and is given by the formula w=âPÎV, where w is the work, P is the external pressure, and ÎV is the change in volume. If the volume change and pressure are in liters and atmospheres respectively, then the work will have units of liter-atmospheres, which can be converted to joules using the conversion factor 1 Lâ atm=101.3 J. Work can also be expressed as force multiplied by distance: w=FÃd, where w is the work in joules, F is the force in newtons, and d is the distance in meters. Note that 1 Nâ m=1 J. | Part A A piston has an external pressure of 9.00 atm. How much work has been done in joules if the cylinder goes from a volume of 0.160 liters to 0.550 liters? Express your answer with the appropriate units. Hints
|