CHEM 130 Chapter Notes - Chapter 3.1-3.2: Covalent Bond, Structural Formula, Chemical Formula

Friday, February 8, 2019
CHEM READING NOTES unit 2
Chapter 3
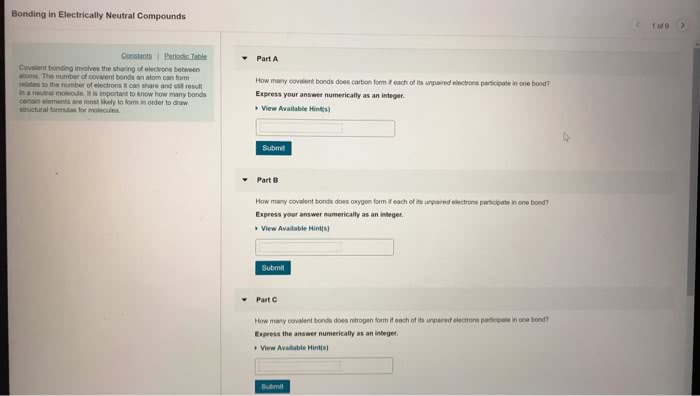
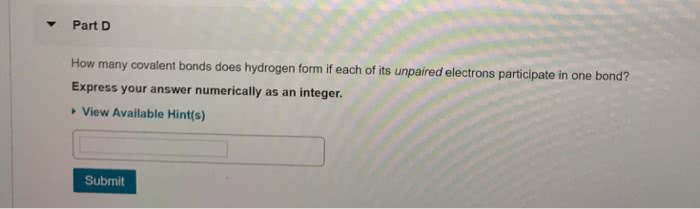
3.1 Types of Chemical Bonds
• John Dalton was first to recognize that chemical compounds are collections of atoms, but he
could not determine the structure of atoms or their means for binding each other
• We later learn atoms have electrons that participate in bonding one atom to another
•
• Forces that hold atoms together in compounds are called chemical bonds
•Chemical bonds: the force or, more accurately, the energy that holds two atoms together
in a compound
• Atoms form by sharing electrons, covalent bonds, resulting in a collection of atoms called
molecules
•covalent bond: a type of bonding in which electrons are shared by atoms.
•molecules: a bonded collection of two or more atoms of the same or different elements.
• Molecules can be repressed in different ways, the simplest method being the chemical
formula, ex CO2
•Chemical formula: the representation of a molecule in which the symbols for the elements are
used to indicate the types of atoms present and subscripts are used to show the relative
numbers of atoms.
• Example of muleucles that contain covalent bond are Hydrogen (H2), water (H2O), oxygen (O2),
ammonia (NH3), methane (CH4)
• More info is given by the structural formula: the representation of a molecule in which the
relative positions of the atoms are shown and the bonds are indicated by lines.
• Ex. Water H—O—H can be other shapes too
•
Methane models
A) structural formula for
methane
B) space-filling model of
methane (a model of a
molecule showing the
relative sizes of the atoms
and their relative
orientations)
c) ball and stick model (a
molecular model that
distorts the sizes of atoms
but shows bond
relationships clearly.
1
Document Summary
To determine the charge distribution in a molecule, we can study its behavior in an electric eld. Ionic bonding: the electrostatic attraction between oppositely charged ions. Ionic substances are formed when an atom that loses electrons relatively easily reacts with an atom that has a high af nity for electrons. Ionic compounds: a compound that results when a metal reacts with a nonmetal to form a cation and an anion. Example: solid sodium chloride the distance between the centers of the na+ and cl- ions is 2. 76 (0. 276 nm), and ionic energy per pair of ions is. That is, the ion pair has lower energy than the separated ions: coulomb"s law also can be used to calculate the repulsive energy when two like-charged ions are brought together. In this case the calculated value of the energy will have a positive sign.