CHEM 6C Chapter Notes - Chapter 22: Ionic Compound, Reaction Quotient, Equilibrium Constant
64 views4 pages
17 Apr 2016
School
Department
Course
Professor
Document Summary
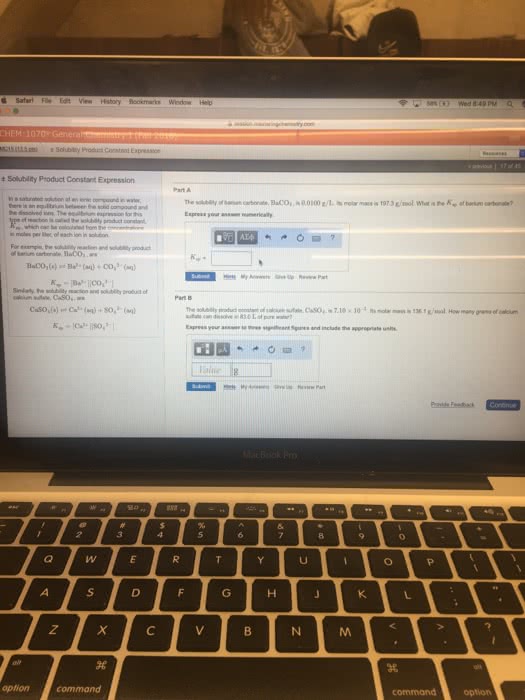
22. 2 common ion efect: the solubility of an ionic solid decreases when a common ion is present in the soluion, ex: determine the solubility of agbro3 in 0. 10m nabro3 soluion. Present in the soluion, so the addiion of the ion shits equilibrium to the let towards the solid agbro3, decreasing its solubility. S = 5. 4 x 10-4m (the solubility in pure water is 7. 3 x 10-3m: common ion efect oten used in separaions in qualitaive analysis. 22. 3 formaion of complexes: the solubility of an ionic solid is increased by the formaion of a soluble complex ion, complex ion: a metal ion with small molecules or ions atached to it. Ex: ag+ + 2nh3 [ag(nh3)2]+ (complexaion reacion) To nh3: agbro3 + 2nh3 [ag(nh3)2]+ + bro3: add agbro3. By adding 2 reacions together, you muliply the k values to get a larger.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232