CHEM 1128Q Chapter Notes - Chapter 13: Endothermic Process, Exothermic Reaction, Reversible Reaction

19
CHEM 1128Q Full Course Notes
Verified Note
19 documents
Document Summary
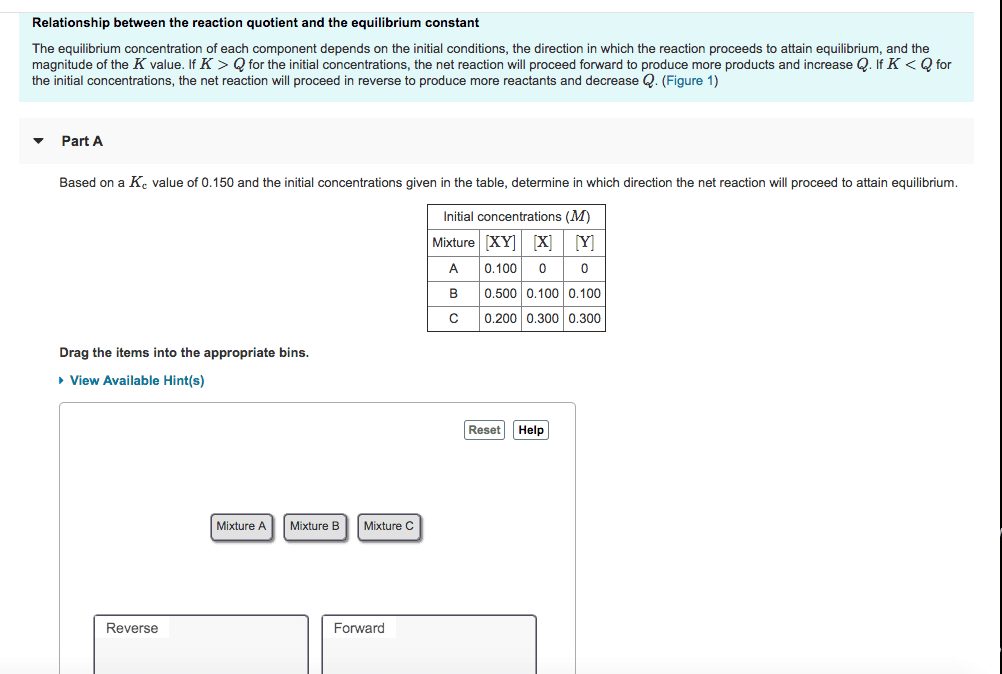
If there are only reactants at the start of a reaction, then qc is initially = 0: qc increases as this reaction proceeds b/c products begin to form and there are less reactants. Increase in temp increase in products reaction shifts to left: decrease in temp increase in reactants reaction shifts to right, endothermic reaction: heat induces the forward reaction process. Increase in temp rxn shifts to the right: decrease in temp rxn shifts to the left, catalysts have no effect on equilibrium, they merely speed up the rate of rxn. Then solve: ice chart is how all of these calculations are done, initial concentrations. In many cases, the reaction starts out with just products or just reactants. The initial concentrations for the components that start of with nothing will be 0: change, when the reaction actually takes place, there will be a shift in formation of products/reactants, equilibrium concentrations.