CHEM 1127Q Chapter Notes - Chapter 4: Chemical Equation, Spectator Ion, Solution

44
CHEM 1127Q Full Course Notes
Verified Note
44 documents
Document Summary
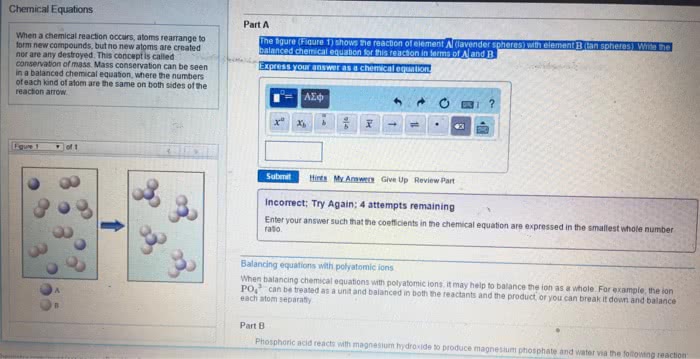
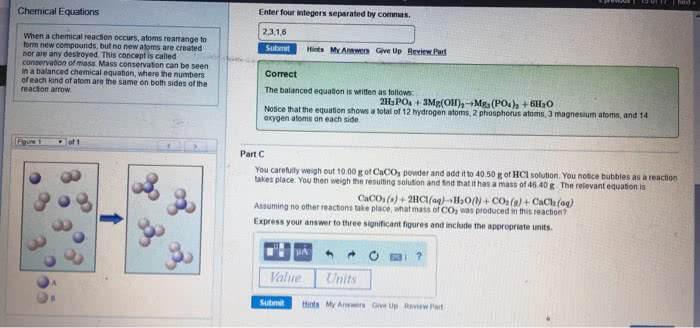
Chemical equations - represents the identities and relative quantities of substance undergoing a chemical/physical change. Plus signs separate individual reactants and products. An arrow separates the reactant (left) and product (right) sides of the equation. Coefficients show the relative numbers of reactants and products (without noting a coefficient of 1) Balances equation - equal number of atoms of each element on the reactant and product side (to satisfy the law of conservation of matter) Change only the coefficients to balance, changing subscripts would alter the substance"s identity. Sum the number of atoms on each side and compare if they are equal. Use fractions instead of integers as intermediate coefficients in the process of balancing a chemical equation. Then be multiplied by a whole number to convert the fractional coefficients to integers without upsetting the balance. Parenthetical abbreviations as subscript show physical state of reactant/product. Solid (s), liquid (l), gas (g), aqueous solutions/substances dissolved in water (aq)