CHEM 1127Q Chapter Notes - Chapter 4: Gravimetric Analysis, Titration, Equivalence Point

44
CHEM 1127Q Full Course Notes
Verified Note
44 documents
Document Summary
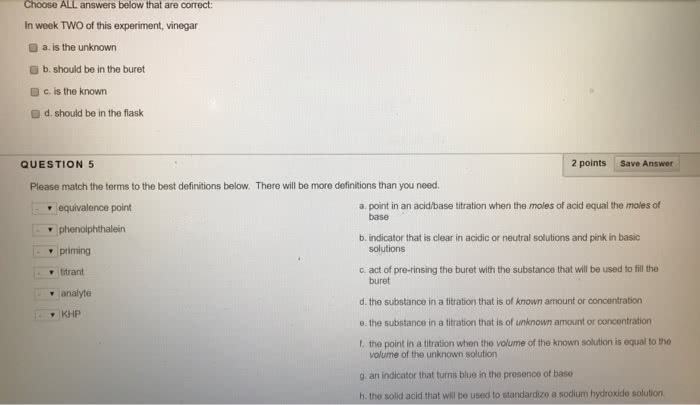
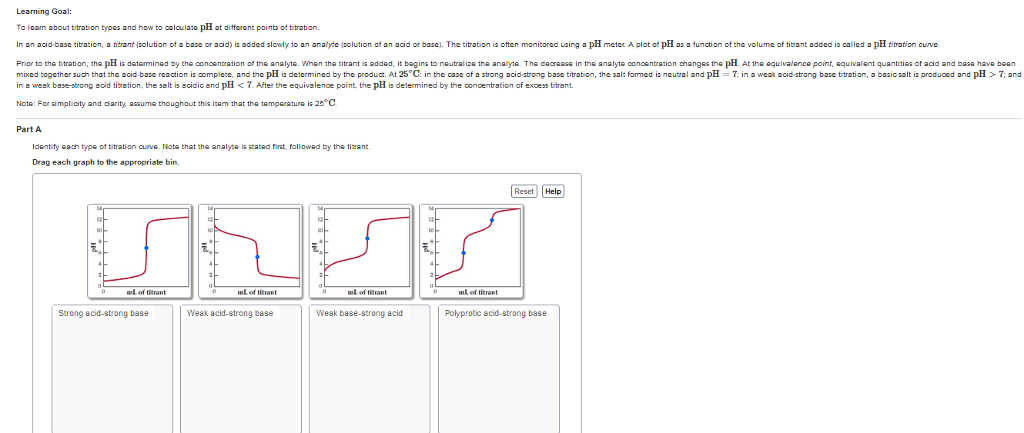
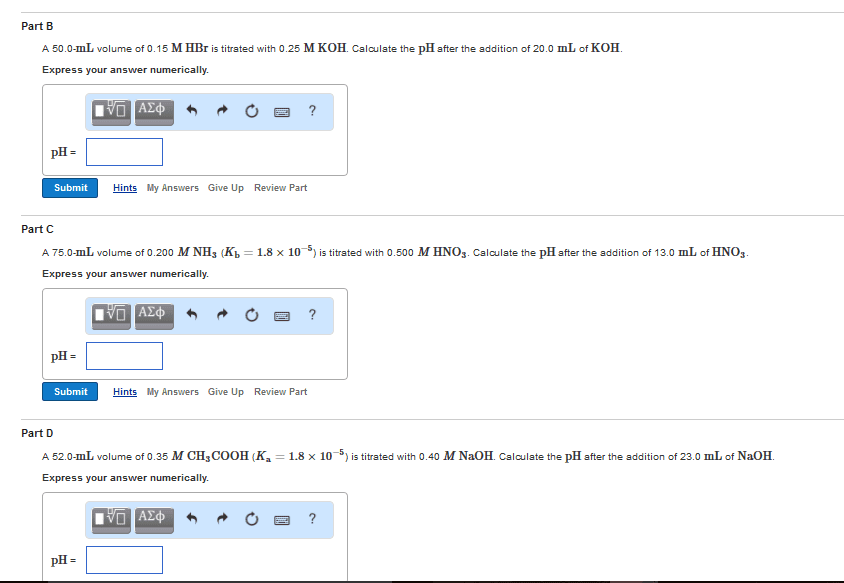
Quantitative analysis - the determination of the amount or concentration of a substance in a sample. A concentration of solute can be determined from the amount reactant that combined with the solute present in a known volume of the solution. The amount of substance present in a sample can be determined by measuring the amount of product that results. Buret - tool used to make incremental additions of a solution containing a known concentration of some substance. Titrant - some substance as mentioned above. Analyte - sample solution containing the substance whose concentration is to be measured. Equivalence point - point of complete reaction. Indicators - to indicate the end of the complete reaction. End point - volume of titrant actually measured. Gravimetric analysis - sample is subjected to some treatment that causes a change in the physical state of the analyte that permits its separation from the other components of the sample.