CHEM 100 Chapter 4-6: Notes
Document Summary
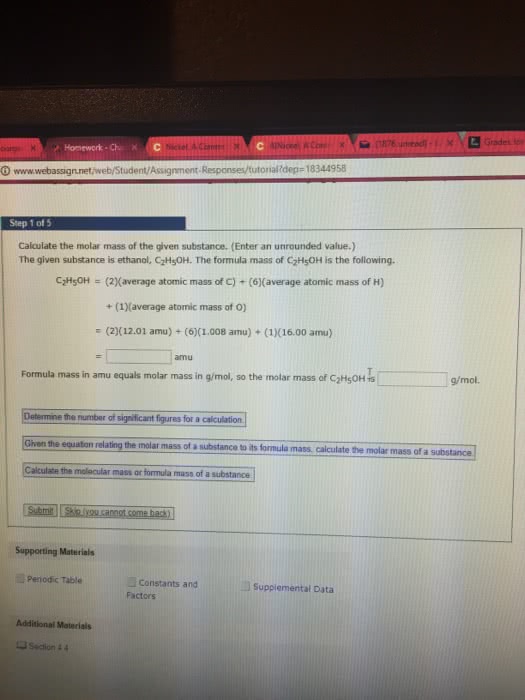
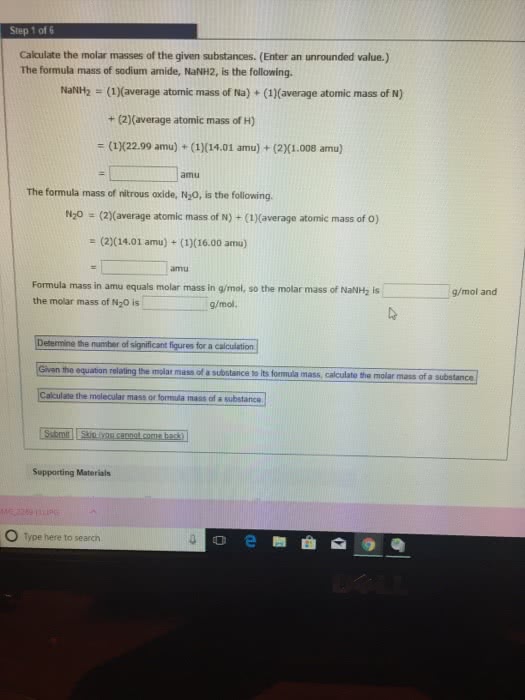
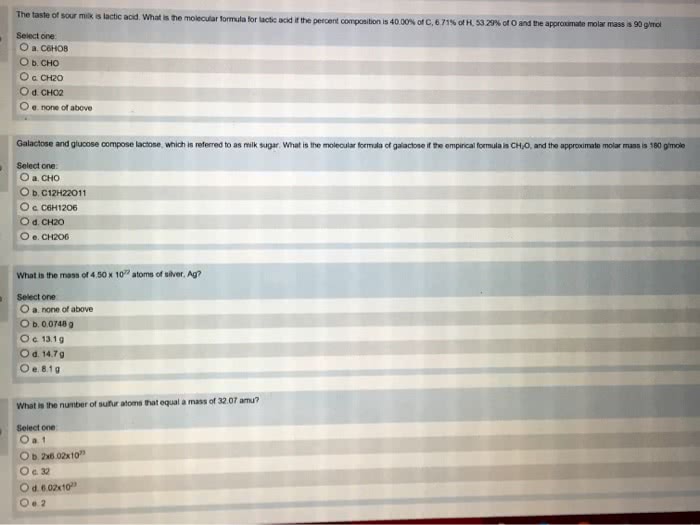
States how many atoms of each element are present in a fundamental unit of a. The sum of the relative masses of all the atoms in a formula unit. The mass in amu of a molecule of a molecular substance. Not possible to weigh out one atom of an element. Can weigh out a mass equal to its relative mass. Can do the same thing with a compound. Ex: 180. 2 amu of c6h12o6 180. 2 g of c6h12o6. Number of atoms or molecules contained in a sample that has a mass equal to its. The mass in grams of 1 mole of a compound. Find the # of moles of c6h12o6 in 27. 0g of c6h12o6. 1. 0g c6h12o6: 3. 35 x 10 molecules. The simplest whole number ratio of atoms in a compound. Assume 100 g of a substance and express the mass of each element in grams. Divide each by the smallest number of moles.