CHEM 1201 Chapter : 3 20 17 Exam 3 Review
Document Summary
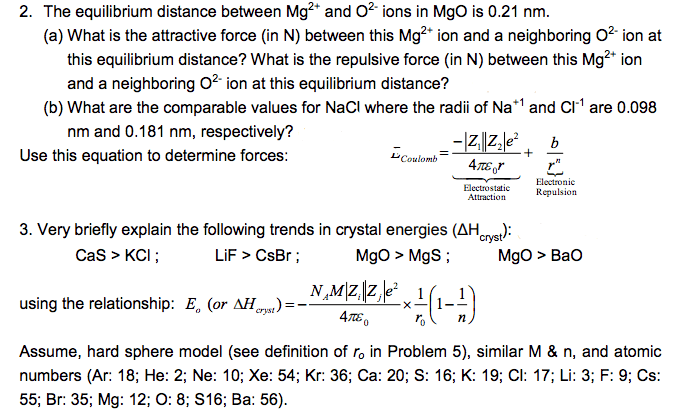
Session times: sundays 6:00 p. m. 7:30 p. m. in 141- c middleton library. Thursdays 6:00 p. m. 7:30 p. m. in 141-c middleton library. Office hours: wednesdays 1:30 p. m. 3:30 p. m. in allen 39 (basement) #1 which of the following sets of ions/elements in an isoelectronic series: na, mg, ne, na+, mg2+, na, na+, mg2+, ne, s2-, cl-, ne, s2-, cl-, ca+ (g: o2(g) + e- o2, o (g) + e- o- (g, o2(g) o2, o (g) o- (g) + e, o+ (g) o (g) + e- Na , na+, ar , f-, br-, se- #4 how many of the following elements/ions have 8 valence electrons: 0, 1, 2, 3, 4, 5. #5 what is the valence electron configuration for mo: [kr]5s24d4, [kr]5s14d5, [kr]4d45s2, [kr]4d55s1, [kr]4d6. #6 what is the oxidation state of cu: +1, +2, -1, -2, can have more than 1. #8 how many valence electrons are in potassium perchlorate: 32, 8, 16, 20, 26.