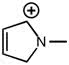
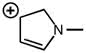
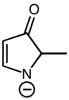
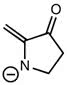
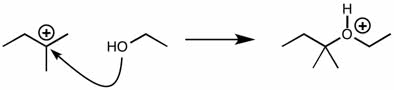
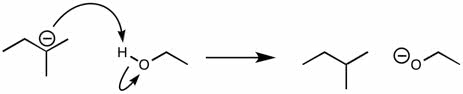
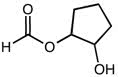
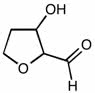
CHEM 213 Chapter Notes - Chapter 14.5-14.7: Molecular Orbital Theory, Pi Bond, Benzene
Document Summary
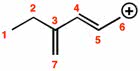
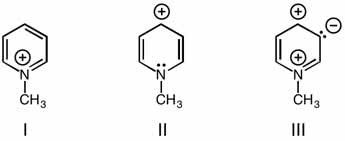
Benzene is very stable: more stable then it was hypothesized to be, this is due to the resonance energy of benzene. Chapter 14. 6: modern theories of the structure of benzene. Quantum mechanics allow for two ways of viewing bonds in molecules: resonance theory, molecular orbital theory. Resonance and the structure of benzene: kekul "s structures i and ii are resonance contributors, not equilibrium forms like he had hypothesized. Things are always more stable in their resonant form. Molecular orbitals and the structure of benzene: carbons in benzene are sp2 and 120 , there are three bonding pi orbitals in benzene. The higher energy set of pi have two nodal planes. The highest pi has three nodal planes. Crystalline benzene involved perpendicular interactions between benzene rings. Huckel"s rule compounds containing one planar ring in which each atom has a p orbital like in benzene: planar monocyclic rings contain 4n+2 electrons.