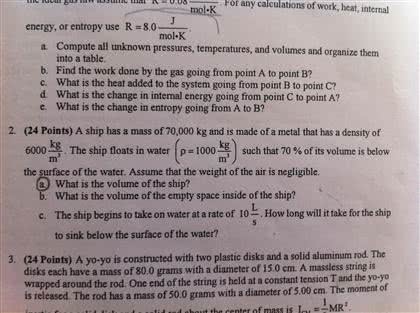PH 122 Chapter Notes - Chapter 19: General Idea, Mean Free Path, Gas Constant
Document Summary
A gas consists of atoms that ll their container"s volume and exert pressure on the container"s walls. There are three macroscopic variables that we use to char- acterize the mechanics of a gas from a motion standpoint: volume, denoted by v , pressure, denoted by p, temperature, denoted by t . The kinetic theory of gases relates the motion of gas atoms to its volume, pressure, and temperature. A mole is the number of atoms in a 12 grams sample of carbon-12. The avogadro"s number - denoted na - is the number of atoms or molecules contained in a mole. Now, we can determine the number of moles n in a sample of any substance as: n = Na (2) where n is the number of moles of gas and r is the gas constant de ned as: The ideal gas law can be rewritten using the boltzmann constant k: (5) where pv = n kt k =