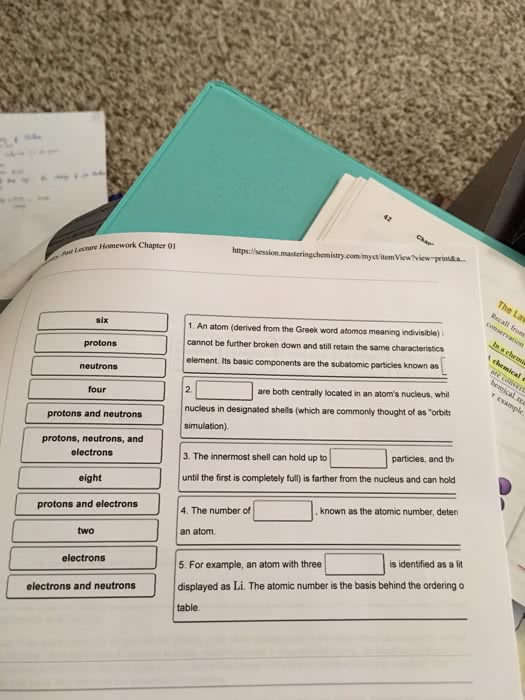
1. (1 point) What might be damaged by ionizing radiation? * paper soft tissue wood lead
2. (1 point) When an atom emits an beta particle, what is the change in the atomic number? * no change decrease by 1 decrease by 2 increase by 1 increase by 2
3. (1 point) What is the change in atomic mass caused by the emission of gamma radiation? * no change decrease by 1 decrease by 2 increase by 1 increase by 2
4. (1 point) Which of the following materials would effectively stop gamma radiation? * one foot of concrete single sheet of paper thin pieces of wood three inches of lead
5. (1 point) When the ratio of neutron-to-proton dictates that a neutron must break down it will form _________. * A helium nucleus An alpha particle A proton and an electron Two protons
6. (1 point) What determines the type of decay if a nucleus falls outside the band of stability? * the ratio of alpha particles to beta particles the ratio of beta particles to alpha particles the ratio of neutrons to protons the ratio of protons to neutrons
7. (1 point) A positron is emitted when ____________. * an alpha particle forms a beta particle forms proton converts to a neutron neutron converts to a proton
8. (1 point) How does an atom with too few neutrons relative to protons undergo radioactive decay? * by converting proton to a neutron by converting a neutron to a proton by emitting an alpha particle by emitting a beta particle
9. (1 point) How much of the original sample would remain after two half lives have past? * There would be nothing of the original left There would be half of the original sample still there There would be one quarter of the sample still present There would only be one eighth of the original sample still present
10. (1 point) What is the approximate half life of carbon- 14? * hundreds of years thousands of years million of years billion of years
11. (1 point) A reaction where a heavier nucleus splits into smaller nuclei is called ___________. * a chemical reaction background radiation fission fusion
12. (1 point) __________ and ___________ are used as moderators to capture neutrons in a nuclear reactor. * Cadmium and other metal Carbon and water Liquid sodium and water Plutonium and neptunium
13. (1 point) For a person working in a place where they might be exposed to radiation a _________ is routinely used to check their exposure. * film badge Geiger counter moderating rod scintillation counter
14. (1 point) Radioisotopes are used to do all of the following except __________. * analyze matter. diagnose medical problems. improve the flavor of food. treat diseases. study plant growth. Open response questions: Answer the following with complete sentences or if math is involve show the work
15. (3 point) The radioisotope radon-222 has a half-life of 3.8 days. How much of a 198.6 g sample of radon -222 would be left after approximately 23 days? ( Show your work) * Your answer
16. (3 point) How can an element be artificially transmuted? Give an example that has an element and what it is transmuted into. * Your answer
17. (4 point) Why does the mass and charge of an alpha particle make it less penetrating than a beta or gamma? Discuss both the mass and the charge(2 points each). *