14
answers
1
watching
601
views
23 Feb 2024
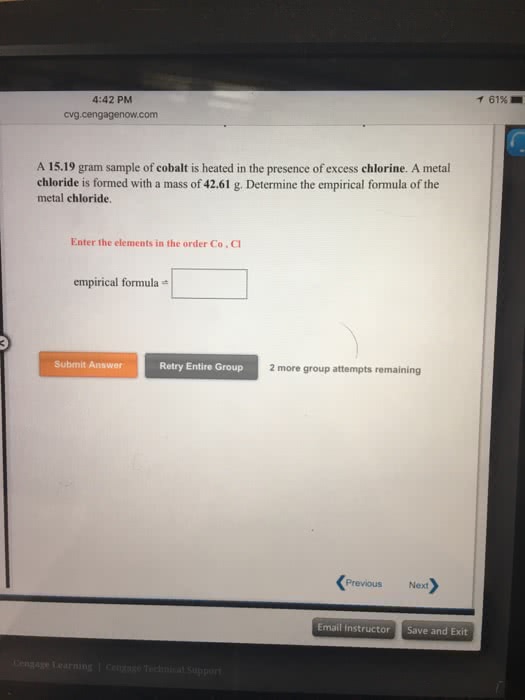
A 27.67 gram sample of iron is heated in the presence of excess chlorine. A metal chloride is formed with a mass of 62.79 g. Determine the empirical formula of the metal chloride.
A 27.67 gram sample of iron is heated in the presence of excess chlorine. A metal chloride is formed with a mass of 62.79 g. Determine the empirical formula of the metal chloride.
Read by 1 person
4 Mar 2024
Already have an account? Log in
Read by 1 person
wigglywiggLv4
28 Feb 2024
Already have an account? Log in
Read by 1 person
Read by 1 person
Read by 1 person
Read by 1 person
adaxia0226Lv1
26 Feb 2024
Already have an account? Log in
Read by 1 person
profsolo88Lv3
25 Feb 2024
Already have an account? Log in
Read by 2 people
Read by 3 people
Read by 3 people
24 Feb 2024
Already have an account? Log in
Read by 3 people
24 Feb 2024
Already have an account? Log in
Read by 3 people
habtyafe231Lv2
24 Feb 2024
Already have an account? Log in