1
answer
2
watching
102
views
cmall98Lv1
9 Jun 2023
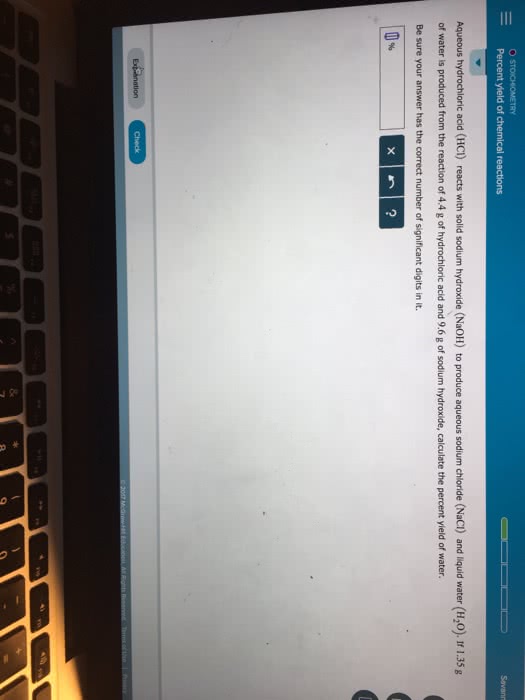
Aqueous hydrochloric acid (HCl) will react with solid sodium hydroxide NaOH to produce aqueous sodium chloride (NaCl) and liquid water (H2O). Suppose 2.9 g of hydrochloric acid is mixed with 6.11 g of sodium hydroxide. Calculate the maximum mass of water that could be produced by the chemical reaction. Round your answer to 2 significant digits.
Aqueous hydrochloric acid (HCl) will react with solid sodium hydroxide NaOH to produce aqueous sodium chloride (NaCl) and liquid water (H2O). Suppose 2.9 g of hydrochloric acid is mixed with 6.11 g of sodium hydroxide. Calculate the maximum mass of water that could be produced by the chemical reaction. Round your answer to 2 significant digits.
Liked by cmall98 and 1 others
9 Jun 2023