5
answers
1
watching
248
views
armaniasmithLv1
18 Oct 2022
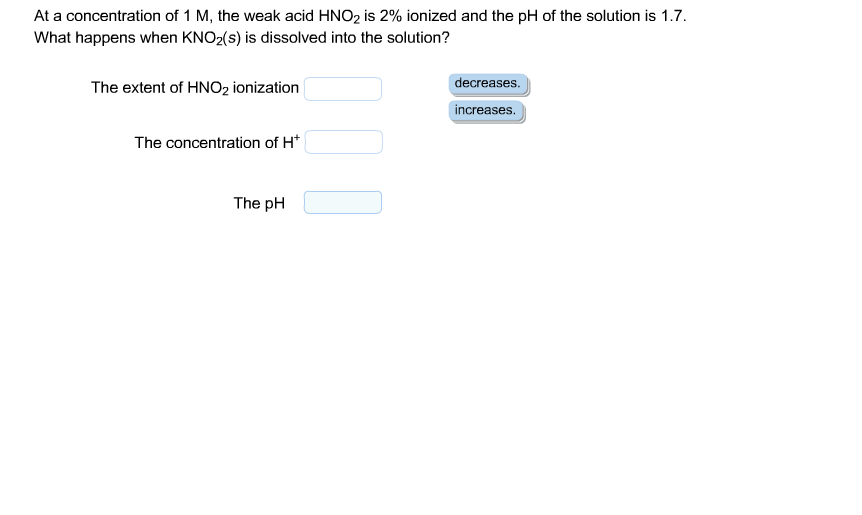
At a concentration of 1 M, the weak acid HNO2 is 2% ionized, and the pH of the solution is 1.7.
What happens when KNO2(s) is dissolved into the solution?
At a concentration of 1 M, the weak acid HNO2 is 2% ionized, and the pH of the solution is 1.7.
What happens when KNO2(s) is dissolved into the solution?
wahabmunir796Lv10
15 Jul 2023
Already have an account? Log in
prathugayamLv8
21 Oct 2022
Already have an account? Log in
Read by 1 person
Read by 1 person
18 Oct 2022
Already have an account? Log in