2
answers
0
watching
23
views
13 Dec 2019
The following values may be useful when solving thistutorial.
Constant Value E?Cu 0.337 V E?Fe -0.440V R 8.314 J?mol?1?K?1 F 96,485 C/mol T 298 K
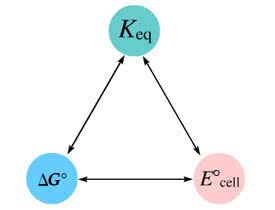
In the activity, click on the E?cell and Keqquantities to observe how they are related. Use this relation tocalculate Keq for the following redox reaction that occursin an electrochemical cell having two electrodes: a cathode and ananode. The two half-reactions that occur in the cell are
Cu2+(aq)+2e??Cu(s) and Fe(s)?Fe2+(aq)+2e?
The net reaction is:
Cu2+(aq)+Fe(s)?Cu(s)+Fe2+(aq)
Use the given standard reduction potentials in your calculationas appropriate.
Express your answer numerically to three significantfigures.
The following values may be useful when solving thistutorial.
| Constant | Value |
| E?Cu | 0.337 V |
| E?Fe | -0.440V |
| R | 8.314 J?mol?1?K?1 |
| F | 96,485 C/mol |
| T | 298 K |
In the activity, click on the E?cell and Keqquantities to observe how they are related. Use this relation tocalculate Keq for the following redox reaction that occursin an electrochemical cell having two electrodes: a cathode and ananode. The two half-reactions that occur in the cell are
Cu2+(aq)+2e??Cu(s) and Fe(s)?Fe2+(aq)+2e?
The net reaction is:
Cu2+(aq)+Fe(s)?Cu(s)+Fe2+(aq)
Use the given standard reduction potentials in your calculationas appropriate.
Express your answer numerically to three significantfigures.
purshuLv2
25 Jun 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Nelly StrackeLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in