2
answers
0
watching
15
views
13 Dec 2019
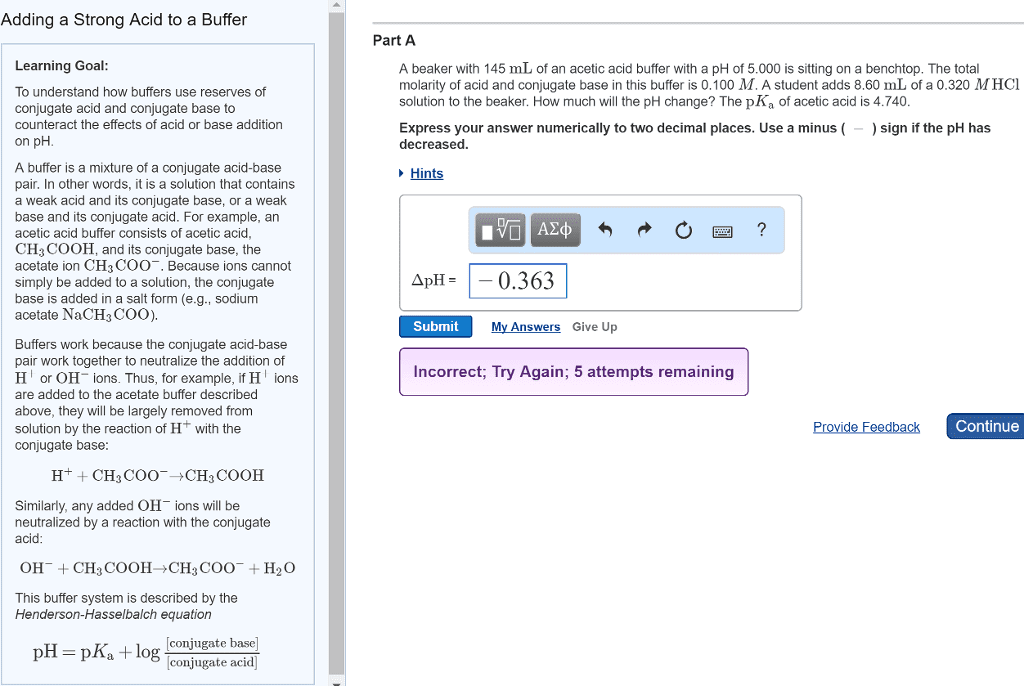
One beaker contains 10.0 mL of acetic acid/sodium acetate buffer at maximum buffer capacity (equal concentrations of acetic acid and sodium acetate), and another contains 10.0 mL of pure water. Calculate the hydronium ion concentration and the pH after the addition of 0.25 mL of 0.10 M HCl to each one. What accounts for the difference in the hydronium ion concentrations? Explain this based on equilibrium concepts; in other words, saying that âone solution is a bufferâ is not sufficient. H3O+(aq) + C2H3O2-(aq) â HC2H3O2(aq) +H2O Ka of acetic acid = 1.8 x 10-5 Acetic acid/sodium acetate buffer: Acetic acid = 0.1M, Sodium acetate = 0.1M
show work
One beaker contains 10.0 mL of acetic acid/sodium acetate buffer at maximum buffer capacity (equal concentrations of acetic acid and sodium acetate), and another contains 10.0 mL of pure water. Calculate the hydronium ion concentration and the pH after the addition of 0.25 mL of 0.10 M HCl to each one. What accounts for the difference in the hydronium ion concentrations? Explain this based on equilibrium concepts; in other words, saying that âone solution is a bufferâ is not sufficient. H3O+(aq) + C2H3O2-(aq) â HC2H3O2(aq) +H2O Ka of acetic acid = 1.8 x 10-5 Acetic acid/sodium acetate buffer: Acetic acid = 0.1M, Sodium acetate = 0.1M
show work
johnjehu68Lv6
25 Jan 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Trinidad TremblayLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in