2
answers
0
watching
11
views
13 Dec 2019
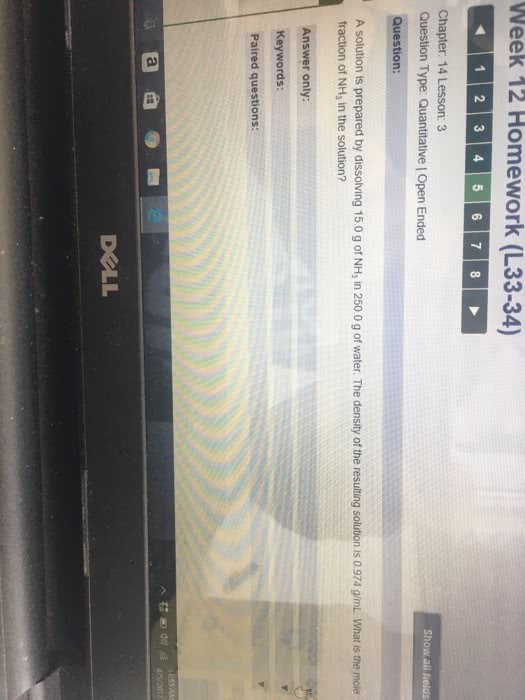
A solution is prepared by dissolving 11.0 g of NH3 in 250.0 g of water. The density of the resulting solution is 0.974 g/mL. The mole fraction of NH3 in the solution is ________.
A. 16.8
B. 0.0440
C. 0.940
D. 0.922
E. 0.0445
A solution is prepared by dissolving 11.0 g of NH3 in 250.0 g of water. The density of the resulting solution is 0.974 g/mL. The mole fraction of NH3 in the solution is ________.
| A. 16.8 | ||
| B. 0.0440 | ||
| C. 0.940 | ||
| D. 0.922 | ||
| E. 0.0445 |
findnoob573Lv10
25 Apr 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Bunny GreenfelderLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in