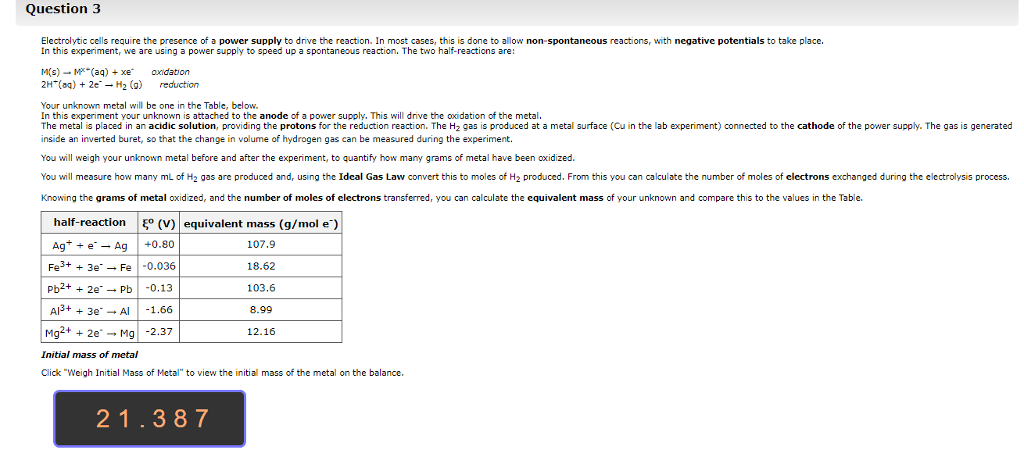
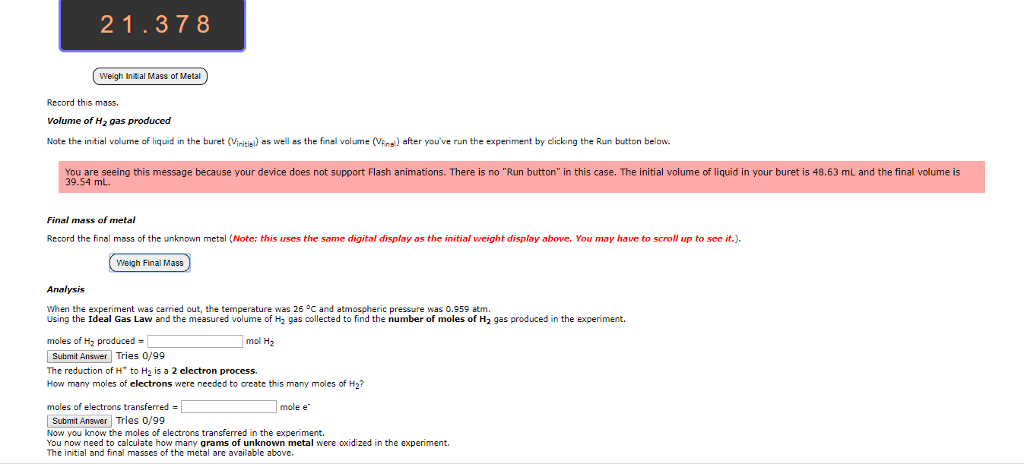
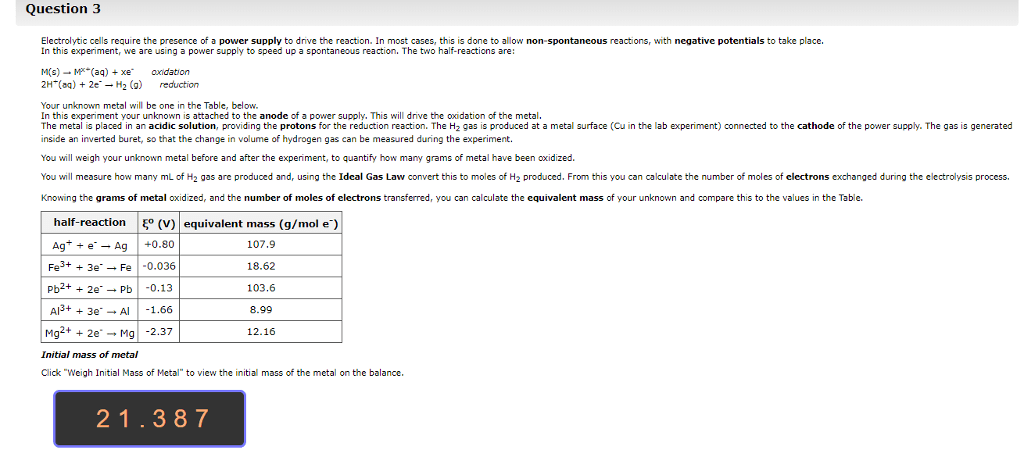
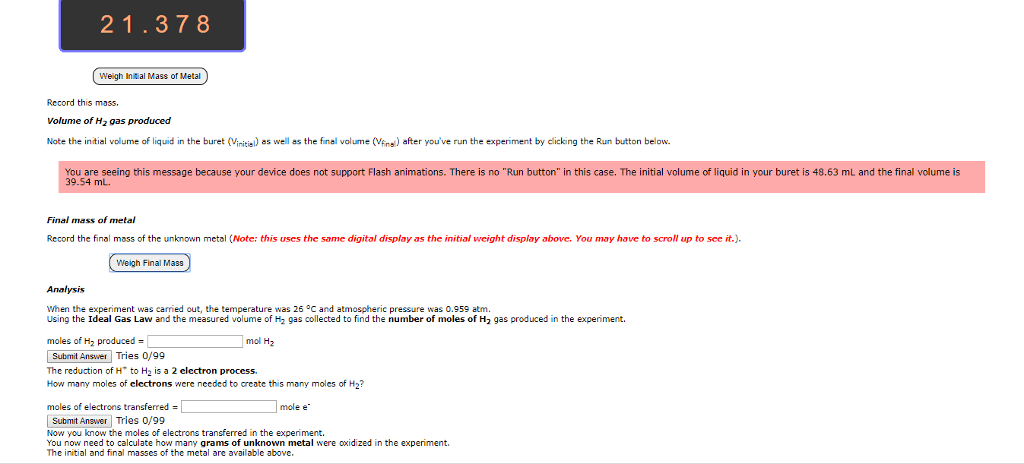
Hello, I would appreciate if someone were to help me answer the following questions. Any help would be much appreciated. Thank you!
1.
2Iâ+Cl2âI2+2Clâ
Which substance is oxidized?
Which substance is reduced?
2.
Oxidation half-reaction for 2Al(s)+6H+(aq)â2Al3+(aq)+3H2(g)
Reduction half-reaction for 2Al(s)+6H+(aq)â2Al3+(aq)+3H2(g)
Oxidation half-reaction for 2Cu+(aq)+Mg(s)âMg2+(aq)+2Cu(s)
Reduction half-reaction for 2Cu+(aq)+Mg(s)âMg2+(aq)+2Cu(s)
3.
Oxidized or Reduced?
Zinc
Water
Iron
Copper
4.
Which of the following increases or decreases the corrosion rate?
Electroplaying the metal surface
Painting the metal surface
Putting strain on the metal
Coating the metals with varnish or grease
Exposing the metal to an electrolyte
Placing the metal objcect in a moist room
5.
Which of these occur at anode vs. at cathode during the rusting of iron?
Oxygen gains electrons
Iron loses electrons
OH- is formed
Fe2+ is formed
6.
What is the main active ingredient in common household bleach, also known as chlorine bleach?
a. Ca(OCI)2
b. H2O2
c. HOCl
d. NaOCl
7.
Oxidation or reduction?
Sn2+ ------> Sn4+ + 2e-
Bi3+ + 3e- -----> Bi
Zn -----> Zn2+ + 2e-
H+ + e- ------> H
8.
Combine the following two reactions to obtain a balanced overall redox reaction.
Sn2+ â Sn4++2eâ and Bi3++3eâ â Bi
Zn â Zn2++2eâ and H++eâ â H
9.
Which of the following statements about freeing metals from their ores is true?
a. Other metals cannot be used to remove a metal from its ore.
b. Vitamin C removes metal from its ore by inhibiting air oxidation.
c. Hydrogen cannot be used to remove metal from its ore because it is an oxidizing agent.
d. Coke or coal can be used to remove a metal from its ore.
Thanks in advance!
Hello, I would appreciate if someone were to help me answer the following questions. Any help would be much appreciated. Thank you!
1.
2Iâ+Cl2âI2+2Clâ
Which substance is oxidized?
Which substance is reduced?
2.
Oxidation half-reaction for 2Al(s)+6H+(aq)â2Al3+(aq)+3H2(g)
Reduction half-reaction for 2Al(s)+6H+(aq)â2Al3+(aq)+3H2(g)
Oxidation half-reaction for 2Cu+(aq)+Mg(s)âMg2+(aq)+2Cu(s)
Reduction half-reaction for 2Cu+(aq)+Mg(s)âMg2+(aq)+2Cu(s)
3.
Oxidized or Reduced?
Zinc
Water
Iron
Copper
4.
Which of the following increases or decreases the corrosion rate?
Electroplaying the metal surface
Painting the metal surface
Putting strain on the metal
Coating the metals with varnish or grease
Exposing the metal to an electrolyte
Placing the metal objcect in a moist room
5.
Which of these occur at anode vs. at cathode during the rusting of iron?
Oxygen gains electrons
Iron loses electrons
OH- is formed
Fe2+ is formed
6.
What is the main active ingredient in common household bleach, also known as chlorine bleach?
a. Ca(OCI)2
b. H2O2
c. HOCl
d. NaOCl
7.
Oxidation or reduction?
Sn2+ ------> Sn4+ + 2e-
Bi3+ + 3e- -----> Bi
Zn -----> Zn2+ + 2e-
H+ + e- ------> H
8.
Combine the following two reactions to obtain a balanced overall redox reaction.
Sn2+ â Sn4++2eâ and Bi3++3eâ â Bi
Zn â Zn2++2eâ and H++eâ â H
9.
Which of the following statements about freeing metals from their ores is true?
a. Other metals cannot be used to remove a metal from its ore.
b. Vitamin C removes metal from its ore by inhibiting air oxidation.
c. Hydrogen cannot be used to remove metal from its ore because it is an oxidizing agent.
d. Coke or coal can be used to remove a metal from its ore.
Thanks in advance!