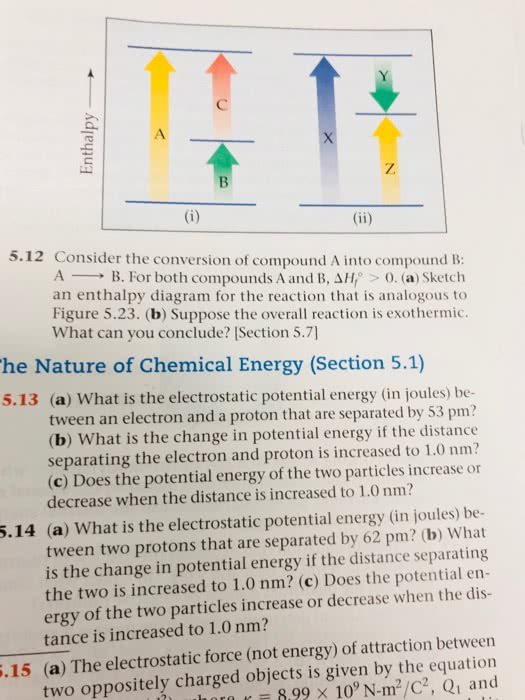
1
answer
0
watching
16
views
13 Dec 2019
what is the difference between plasmon peak positions between the citrate and borate stabilized nanoparticles? How does that correlate to the stated size of the particles?
Base on the rate of nanoparticle formation, what can you conclude about the reducing potential of sodium citrate vs NaBH4?
what is the difference between plasmon peak positions between the citrate and borate stabilized nanoparticles?
How does that correlate to the stated size of the particles?
Base on the rate of nanoparticle formation, what can you conclude about the reducing potential of sodium citrate vs NaBH4?
1
answer
0
watching
16
views
For unlimited access to Homework Help, a Homework+ subscription is required.
22 Feb 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232