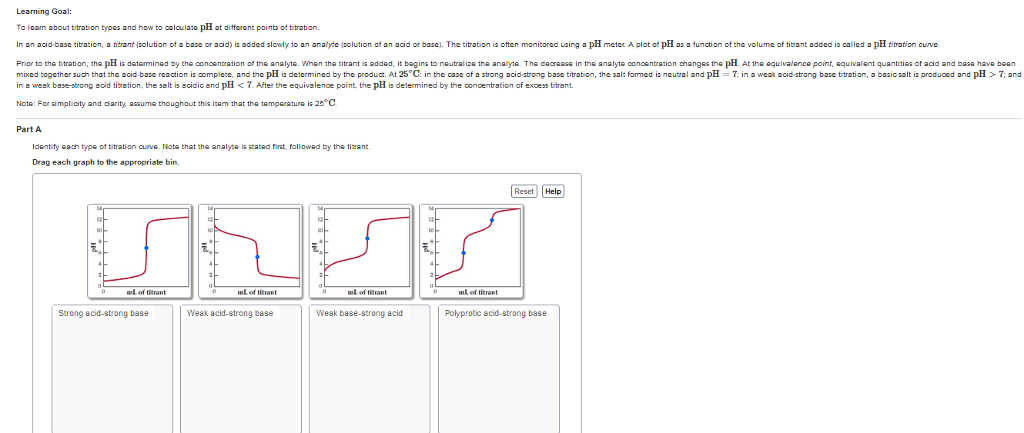
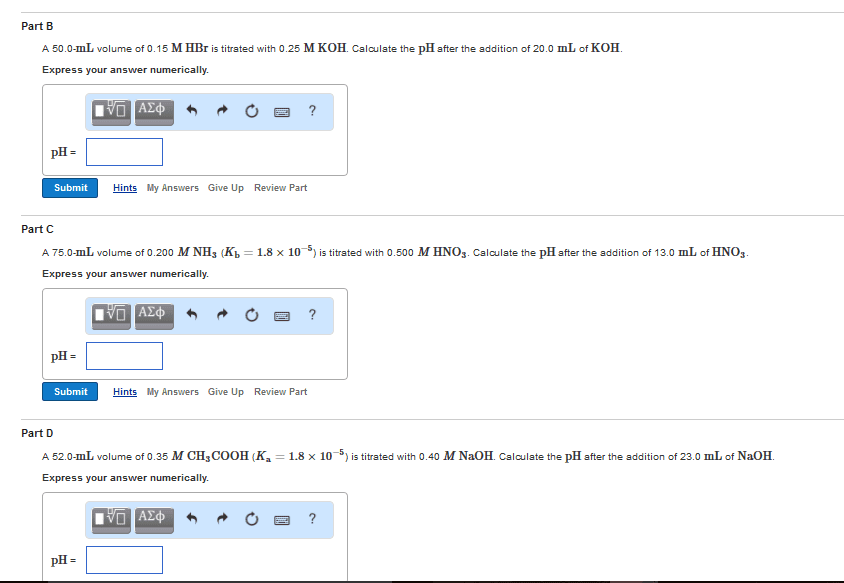
Determine the equivalent acid-base equilibrium that results when each of the following pairs of solutions is mixed. 25.0 mL of 0.50 M NaOH + 12.0 mL of 1.50 M HCl Pick two initial conditions from first four choices. Pick one choice from remaining choices to indicate type of equivalent solution.Choose at least one answer. a. Weak acid b. Strong acid c. Weak base d. Strong base e. pH determined by excess concentration of diluted strong acid (some acid used to neutralize base) f. pH determined by excess concentration of diluted strong base g. pH determined by concentration of diluted strong acid h. pH determined by buffer equation (resulting mixture produces a buffer) i. pH determined by conjugate base concentration (equivalent to equivalence point in titration of weak acid) j. pH determined by excess concentration of diluted strong base (remaining strong base + weak conjugate base = beyond equivalence point in titration of weak acid)
Determine the equivalent acid-base equilibrium that results when each of the following pairs of solutions is mixed. 25.0 mL of 0.50 M NaOH + 12.0 mL of 1.50 M HCl Pick two initial conditions from first four choices. Pick one choice from remaining choices to indicate type of equivalent solution.Choose at least one answer. a. Weak acid b. Strong acid c. Weak base d. Strong base e. pH determined by excess concentration of diluted strong acid (some acid used to neutralize base) f. pH determined by excess concentration of diluted strong base g. pH determined by concentration of diluted strong acid h. pH determined by buffer equation (resulting mixture produces a buffer) i. pH determined by conjugate base concentration (equivalent to equivalence point in titration of weak acid) j. pH determined by excess concentration of diluted strong base (remaining strong base + weak conjugate base = beyond equivalence point in titration of weak acid)