2
answers
0
watching
14
views
23 Nov 2019
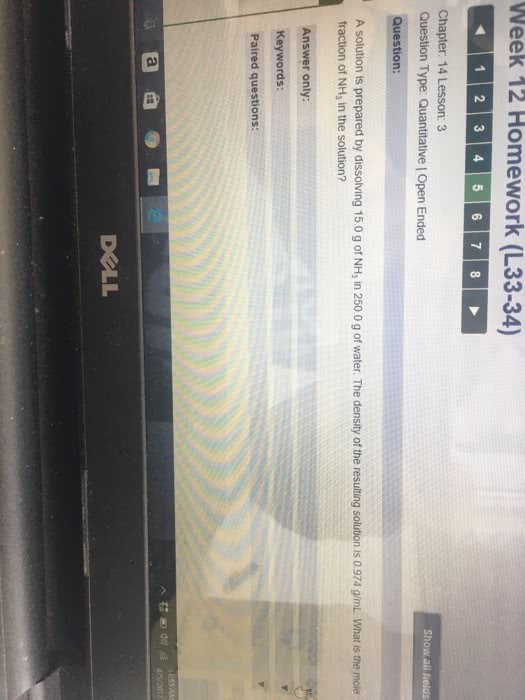
a solution is prepared by dissolving 11.0g of nh3 in 250.0g of water. the density of the resulting solution is 0.974 g/ml. the mole fraction of nh3 in solution is?
0.0445 (answer). How did they get that answer?
a solution is prepared by dissolving 11.0g of nh3 in 250.0g of water. the density of the resulting solution is 0.974 g/ml. the mole fraction of nh3 in solution is?
0.0445 (answer). How did they get that answer?
findnoob573Lv10
25 Apr 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Tod ThielLv2
4 Feb 2019
Get unlimited access
Already have an account? Log in